Johnson & Johnson-sponsored activities at the 21st Congress of ECCO, Stockholm, Sweden, February 18–21, 2026

Date of preparation: March 2026 | EM-173727
Overview: Johnson & Johnson sponsored two satellite symposia at the 21st Congress of the European Crohn’s and Colitis Organisation (ECCO 2026). These symposia were predominantly based on practical and engaging patient case studies that showed how guselkumab clinical trial data can be applied in real-world inflammatory bowel disease (IBD) care. The goal was to give clinicians actionable, evidence-based guidance for IBD management.
Symposium 1
• Title: Mission remission: practical insights for Crohn’s disease in the IL 23 era
• Focus: Crohn's disease (CD)
• Presenters: Anthony Buisson, Axel Dignass, Catherine Reenaers and Stefan Schreiber
• Case types: Adult and elderly patients, across various clinical scenarios
Symposium 2
• Title: Charting the UC horizon: navigating IL 23 in clinical practice
• Focus: Ulcerative colitis (UC)
• Presenters: Ailsa Hart, James Lindsay, Elisabeth Schnoy and Bram Verstockt
• Case types: Biologic naive and biologic experienced patients
Did you miss these symposia? Click on the links below for the full on-demand recordings, and read further for some key highlights from each symposium.
Symposium 1: Mission remission: practical insights for Crohn’s disease in the IL-23 era
Symposium 2: Charting the UC horizon: navigating IL-23 in clinical practice
Symposium 1: Mission remission: practical insights for Crohn’s disease in the IL-23 era
Axel Dignass reviewed 30 years of IBD progress, unmet needs and rising disease prevalence [1], noting that the guselkumab programme aims to shift care from disease control to disease modification. Stefan Schreiber explained the historical perspective of IL-23 and summarised the key differences between the three IL-23 inhibitors: risankizumab, mirikizumab and guselkumab.
Catherine Reenaers, Axel Dignass and Anthony Buisson presented different patient cases covering the age spectrum from early adulthood to middle age and then older age, with each age group presenting its own distinct challenges and goals.
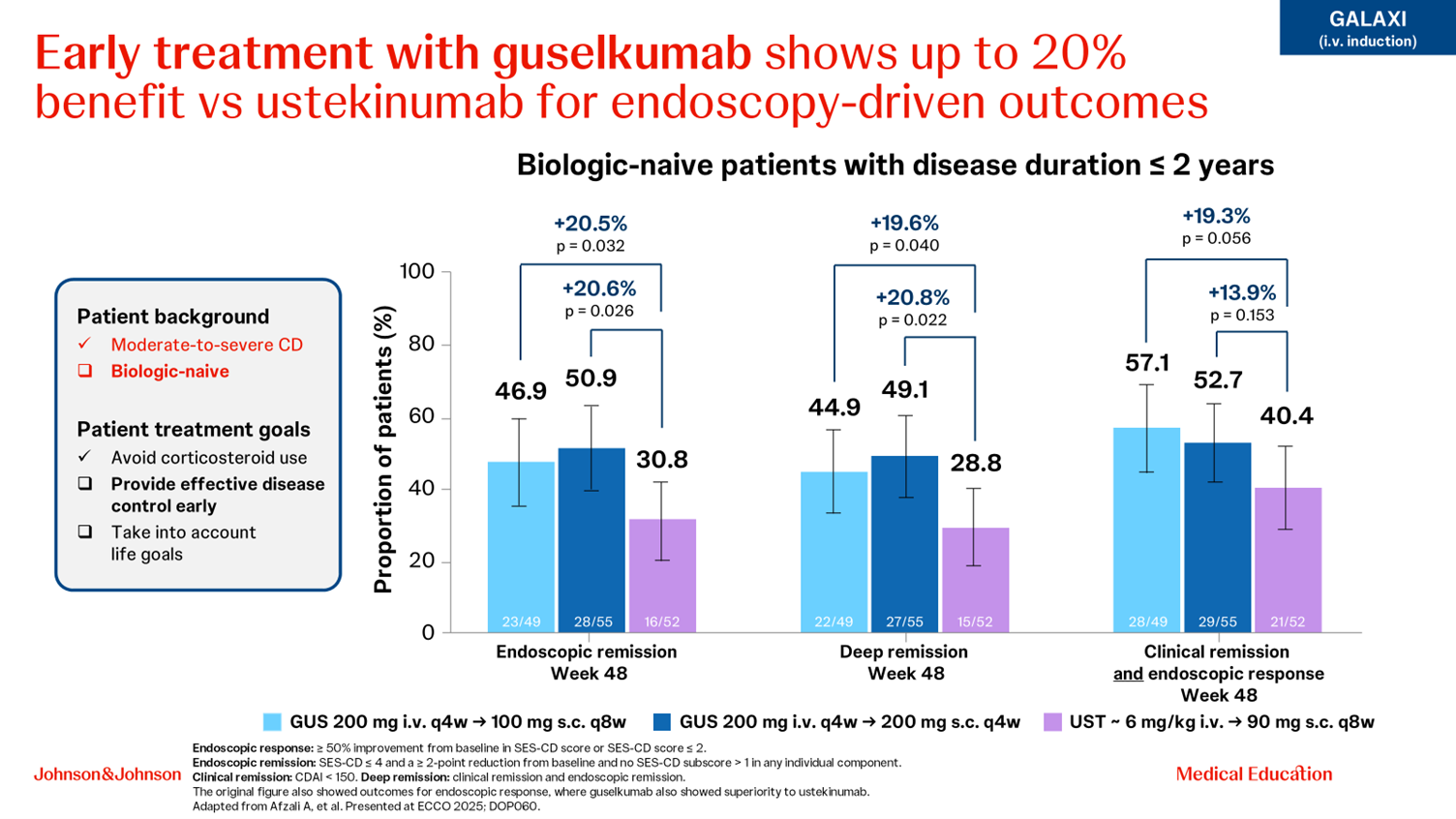
Catherine Reenaers showcased how biologic-naive patients with moderate-to-severe CD need symptomatic relief and long-term disease control with an early, effective therapy, as the benefit may decline with each subsequent treatment [2]. Treatment with guselkumab in biologic-naive patients with a disease duration ≤2 years results in a benefit of up to 20% compared with ustekinumab for endoscopy-driven endpoints [3] (Fig. 1). In the GALAXI study, over 90% of patients in clinical remission at Week 48 also were corticosteroid-free for 90 days [4] and guselkumab showed superiority over ustekinumab in endoscopy-driven endpoints at Week 48 [5].
Figure 1. Data from the GALAXI study showing superiority of guselkumab over ustekinumab in biologic-naive patients with a disease duration of ≤2 years [3].
The second case presented a middle-aged biologic-experienced patient who needed rapid symptomatic relief and sustained long-term outcomes. Axel Dignass shared data from the GRAVITI study which showed fast reduction in abdominal pain and stool frequency within 4 weeks of guselkumab initiation [6], high outcomes in biologic-experienced patients [7], sustained responses for up to 2 years [8] and efficacy in the more difficult-to-treat ileal disease [9].
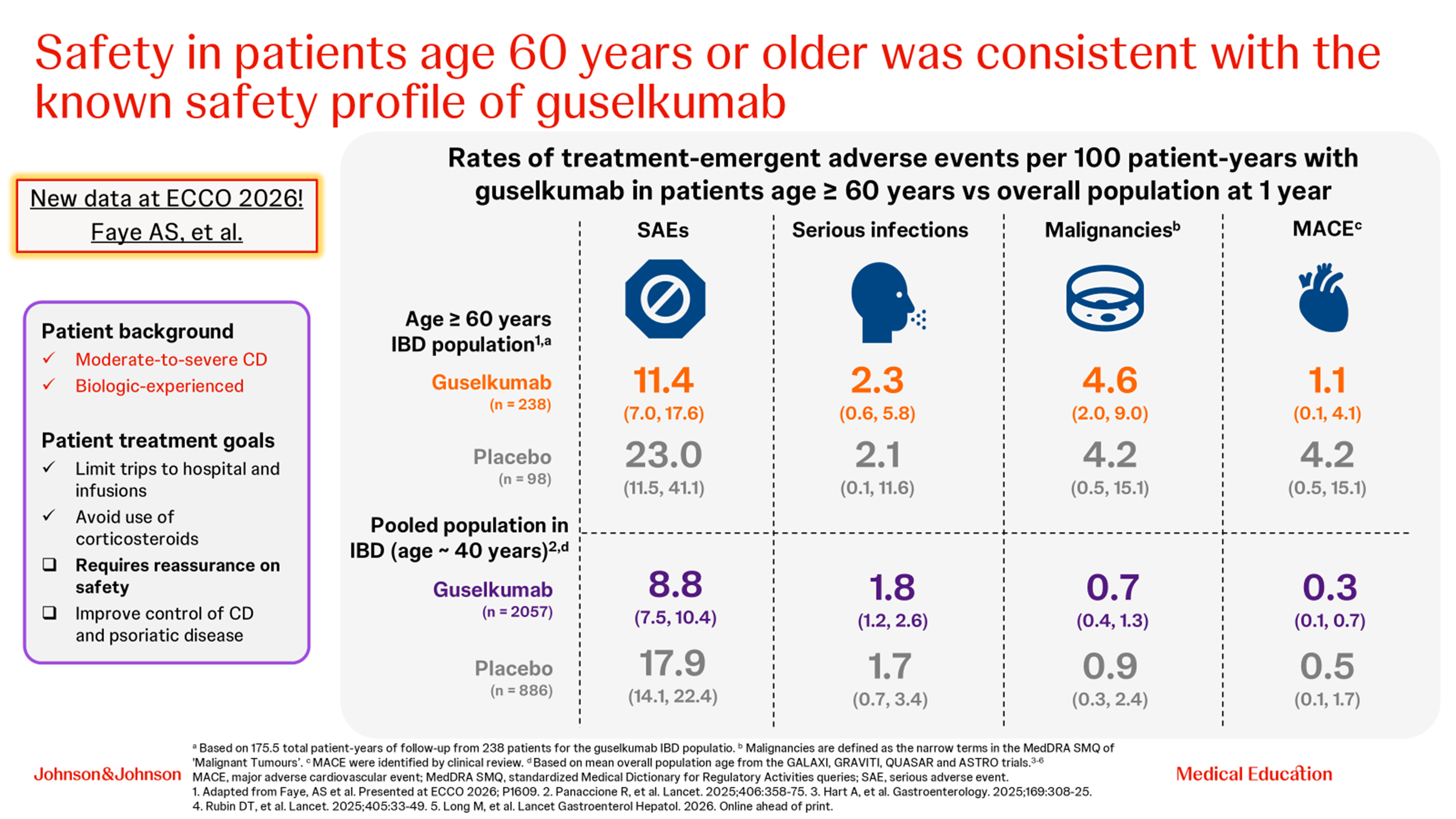
The final case described an older patient with IBD and comorbid psoriasis. Anthony Buisson highlighted that the GRAVITI study of guselkumab has shown high rates of corticosteroid-free clinical remission and endoscopic remission [10], and with a known safety profile [11] (Fig. 2). Guselkumab also helps to resolve arthritis/arthralgia in patients with CD [12] and is the only interleukin (IL)-23 inhibitor that significantly reduced radiographic damage compared with placebo in psoriatic arthritis [13].
Figure 2. Treatment emergent adverse events at 1 year with guselkumab in patients with IBD aged ≥60 years and the pooled IBD population [11].
Finally, Stefan Schreiber discussed how to personalise guselkumab therapy in patients failing ustekinumab [14], patients with a high inflammatory burden [15] and patients who show an inadequate response to guselkumab 100 mg every 8 weeks (q8w) [16].
To conclude, the faculty reiterated the benefits of guselkumab and were awaiting the upcoming results from the FUZION study of guselkumab in fistulising CD.
Symposium 2: Charting the UC horizon: navigating IL-23 in clinical practice
Bram Verstockt described the role of IL-23 in the pathophysiology of IBD and how binding of guselkumab to CD64 enables neutralisation of IL-23 at its cellular source in vitro [17]. Ailsa Hart, James Lindsay, Elisabeth Schnoy and Bram Verstockt then described two patient cases regarding guselkumab use in real-life clinical practice.
The first case study described a patient with UC and previous exposure to biologics. Guselkumab provides significantly better symptomatic response compared with placebo as early as Day 6 [18] and 30% higher clinical remission rates than placebo in patients exposed to one prior antitumour necrosis factor therapy [19].
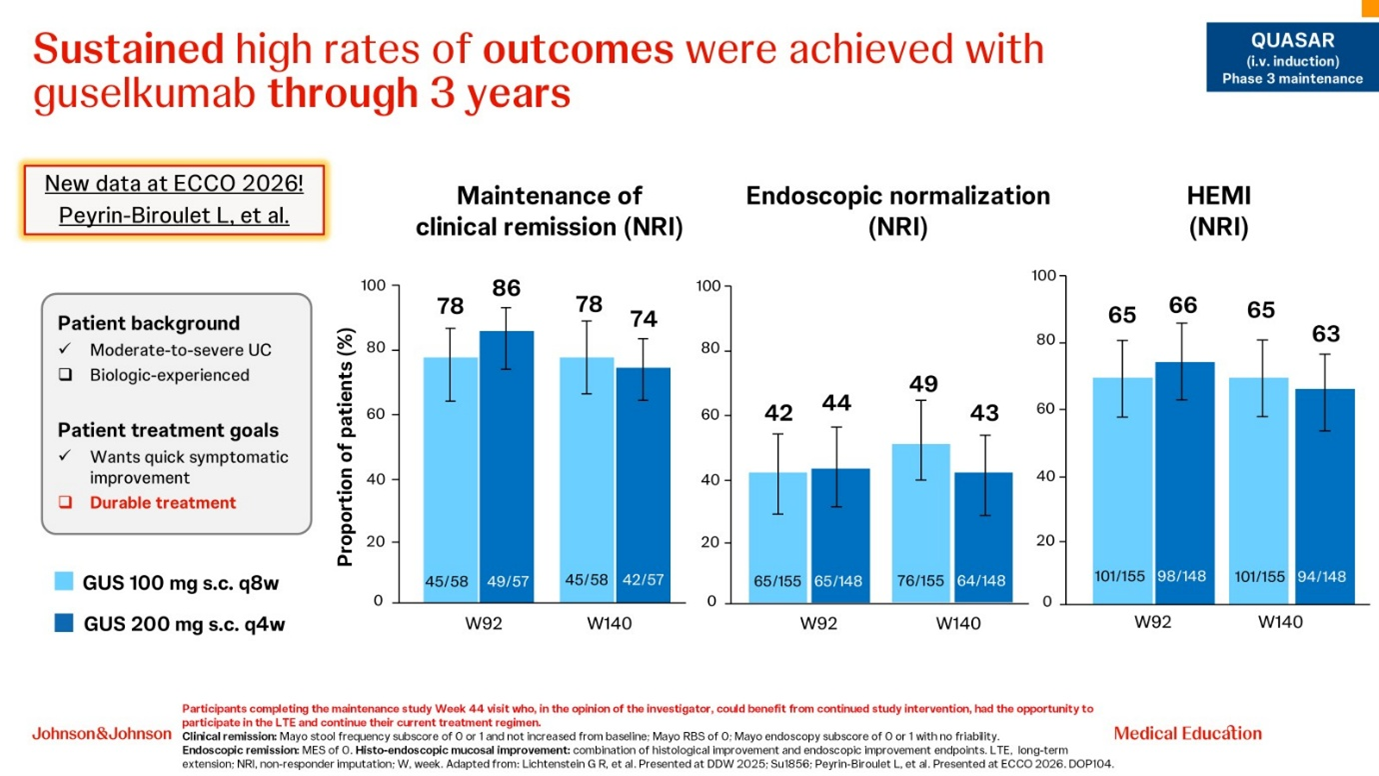
The second case study described the efficacy and safety of guselkumab in a young, female, biologic-naive patient and highlighted that the 3-year long-term extension of QUASAR demonstrated high and sustained rates of clinical remission, endoscopic normalisation and histo-endoscopic mucosal improvement with guselkumab (Fig. 3) [20].
Figure 3. Sustained rates of clinical remission, endoscopic improvement and histo-endoscopic mucosal improvement (HEMI) at Week 92 and Week 140 with guselkumab: long-term extension data from the QUASAR study [20].
This symposium also demonstrated, as shown in symposium 1, how guselkumab is raising the bar for patients with CD and UC and offers the flexibility of both intravenous and subcutaneous induction for the management of patients with IBD [21].
References
- Hracs L, Windsor JW, Gorospe J, et al; Global IBD Visualization of Epidemiology Studies in the 21st Century (GIVES-21) Research Group. Global evolution of inflammatory bowel disease across epidemiologic stages. Nature 2025;642:458–66. doi: 10.1038/s41586-025-08940-0.
- Hanrahan TP, Chan R, Tassone D, et al. Persistence of second and third-line biologics in inflammatory bowel disease: A multi-centre cohort study. Future Pharmacol 2022;2:669–80. doi.org/10.3390/futurepharmacol2040041.
- Afzali A, Rubin DT, Danese S, et al. Early disease efficacy of guselkumab therapy in biologic-naïve patients with moderately to severely active Crohn’s disease: Post-hoc analysis from the phase 3 GALAXI 2 & 3 studies. Presented at ECCO 2025; DOP060.
- Panés J, Hisamats T, Armuzzi A, et al. Corticosteroid sparing effects of treatment with guselkumab in patients with moderately to severely active Crohn’s disease: Phase 3 GALAXI 2/3 results through week 48. Presented at ECCO 2025; P0917.
- Panaccione R, Feagan BG, Afzali A, et al; GALAXI 2 & 3 Study Group. Efficacy and safety of intravenous induction and subcutaneous maintenance therapy with guselkumab for patients with Crohn's disease (GALAXI-2 and GALAXI-3): 48-week results from two phase 3, randomised, placebo and active comparator-controlled, double-blind, triple-dummy trials. Lancet 2025;406:358–75. doi: 10.1016/S0140-6736(25)00681-6.
- Afzali A, Panaccione R, Danese S, et al. Efficacy of guselkumab intravenous and subcutaneous induction: symptoms, heath-related quality of life, and inflammatory biomarker results from the GALAXI and GRAVITI studies. Presented at DDW 2025; Su1853.
- Hart A, Panaccione R, Steinwurz F, et al. Efficacy and safety of guselkumab subcutaneous induction and maintenance in participants with moderately to severely active Crohn's disease: Results from the phase 3 GRAVITI study. Gastroenterology 2025;169:308–25. doi: 10.1053/j.gastro.2025.02.033.
- D’Haens GR, Rubin DT, Afzali A, et al. Efficacy and safety of guselkumab through week 96 after intravenous or subcutaneous induction in participants with Crohn’s disease: Phase 3 long-term extension data from GALAXI 2, GALAXI 3, and GRAVITI. Presented at ACG 2025;P1066.
- Richards D, Seridi L, Sohn K, et al. Endoscopic patient clustering to investigate differential treatment effects of guselkumab and ustekinumab in Crohn’s disease: Post-hoc analysis of GALAXI and GRAVITI trials. Presented at DDW 2025; Su1861.
- Hart A, Panaccione R, Steinwurz F, et al. Corticosteroid sparing effects of treatment with guselkumab in patients with moderately to severely active Crohn’s disease: Phase 3 GRAVITI study results through week 48. Presented at ECCO 2025; P0872.
- Faye AS, Sebastian S, McCaffrey V, et al. Safety of guselkumab in patients aged ≥60 years with immune-mediated inflammatory diseases: a pooled analysis of registrational trials in UC, CD, PsA and PsO. Presented at ECCO 2026;DOP1069.
- Danese S, Panaccione R, Rubin DT, et al, on behalf of the GALAXI-1 investigators. Clinical efficacy and safety of guselkumab maintenance therapy in patients with moderately to severely active Crohn’s Disease: Week 48 analyses from the phase 2 GALAXI 1 study. Presented at ECCO 2026;OP24.
- Kerschbaumer A, Smolen JS, Ferreira RJO, et al. Efficacy and safety of pharmacological treatment of psoriatic arthritis: a systematic literature research informing the 2023 update of the EULAR recommendations for the management of psoriatic arthritis. Ann Rheum Dis 2024;83:760–74. doi: 10.1136/ard-2024-225534.
- Afzali A, Wolf D, Leong RW, et al. Efficacy and safety of subcutaneous guselkumab rescue therapy in patients with moderately to severely active Crohn’s disease and inadequate response to ustekinumab: results from GALAXI 1, 2, & 3 long-term extension. Presented at UEGW 2025; OP005.
- Afzali A, Hisamatsu T, Rubin DT, et al. Guselkumab maintenance dose regimens in patients with high disease activity and severity: subgroup analysis of participants with moderately to severely active Crohn’s disease in the GALAXI Phase 3 Studies. Presented at UEGW 2025; MP582.
- Panaccione R, Hisamatsu T, Afzali A, et al. Efficacy and safety of guselkumab in participants with moderately to severely active Crohn’s disease who had maintenance dose adjustment: Results from the phase 3 GALAXI 2 & 3 long-term extension. Presented at ECCO 2026;P0596.
- Sachen KL, Hammaker D, Sarabia I, et al. Guselkumab binding to CD64+ IL-23-producing myeloid cells enhances potency for neutralizing IL-23 signaling. Front Immunol 2025;16:1532852. doi: 10.3389/fimmu.2025.1532852. eCollection 2025.
- Dignass A, Hirai F, Saruta M, et al. Symptomatic improvement with intravenous guselkumab induction therapy is observed early in patients with moderately to severely active Ulcerative Colitis: post-hoc analysis of QUASAR. Presented at ECCO 2026; OP10.
- Rubin DT, Allegretti JR, Panés J, et al; QUASAR Study Group. Guselkumab in patients with moderately to severely active ulcerative colitis (QUASAR): phase 3 double-blind, randomised, placebo-controlled induction and maintenance studies. Lancet 2025;405:33–49. doi: 10.1016/S0140-6736(24)01927-5.
- Peyrin-Biroulet L, Bressler B, Dignass A et al. Efficacy and safety of guselkumab for ulcerative colitis through week 140 of the QUASAR long-term extension study. Presented at ECCO 2026;DOP104.
- Guselkumab EMA SmPC. Available from: https://www.ema.europa.eu/en/documents/product-information/tremfya-epar-product-information_en.pdf. Accessed March 2026.
Disclaimer: This programme is not affiliated with ECCO.