DOP55 A real-world comparison of the effectiveness and safety of vedolizumab and anti-TNF therapies in early treatment initiation with first-line biologic therapy in ulcerative colitis: Results from EVOLVE
G. Mantzaris1, B. Bressler2, U. Kopylov3, M. Bassel4, N. Brett4, C. Colby5, C. Lopez6, S. Saha7, C. Kifnidi8, C. Agboton9, S. Adsul9, D. Demuth10, M. Luo11, S. Wang11, A. Yarur12
1Department of Gastroenterology, Evangelismos Hospital, Athens, Greece, 2Department of Gastroenterology, St. Paul’s Hospital, Vancouver, Canada, 3Department of Gastroenterology, Sheba Medical Center, Tel Aviv, Israel, 4Evidera, Real-World Evidence, Montreal, Canada, 5Evidera, Real-World Evidence, San Francisco, USA, 6Department of Medical Affairs, Takeda Pharmaceuticals USA Inc., Deerfield, USA, 7Department of Medical Affairs, Takeda Canada Inc., Oakville, Canada, 8Department of Medical Affairs, Takeda Hellas S.A. Athens, Athens, Greece, 9Department of Global Medical Affairs, Takeda Pharmaceuticals International AG, Zurich, Switzerland, 10Evidence Generation and Publications, Takeda Pharmaceuticals International, Singapore, 11Global Outcomes Research, Takeda Pharmaceuticals International, Boston, USA, 12Department of Gastroenterology, Medical College of Wisconsin, Milwaukee, USA
Background
Evidence suggests that early treatment (Tx) with biologic agents in Crohn’s disease improves long-term clinical outcomes. However, there is less evidence in ulcerative colitis (UC), and data comparing early Tx with first-line biologic vedolizumab (VDZ) to anti-tumour necrosis factor (anti-TNF) in real-world settings is needed. This study compared the clinical effectiveness and safety of UC patients who initiated VDZ or an anti-TNF as a first-line biologic within 2 years following diagnosis.
Methods
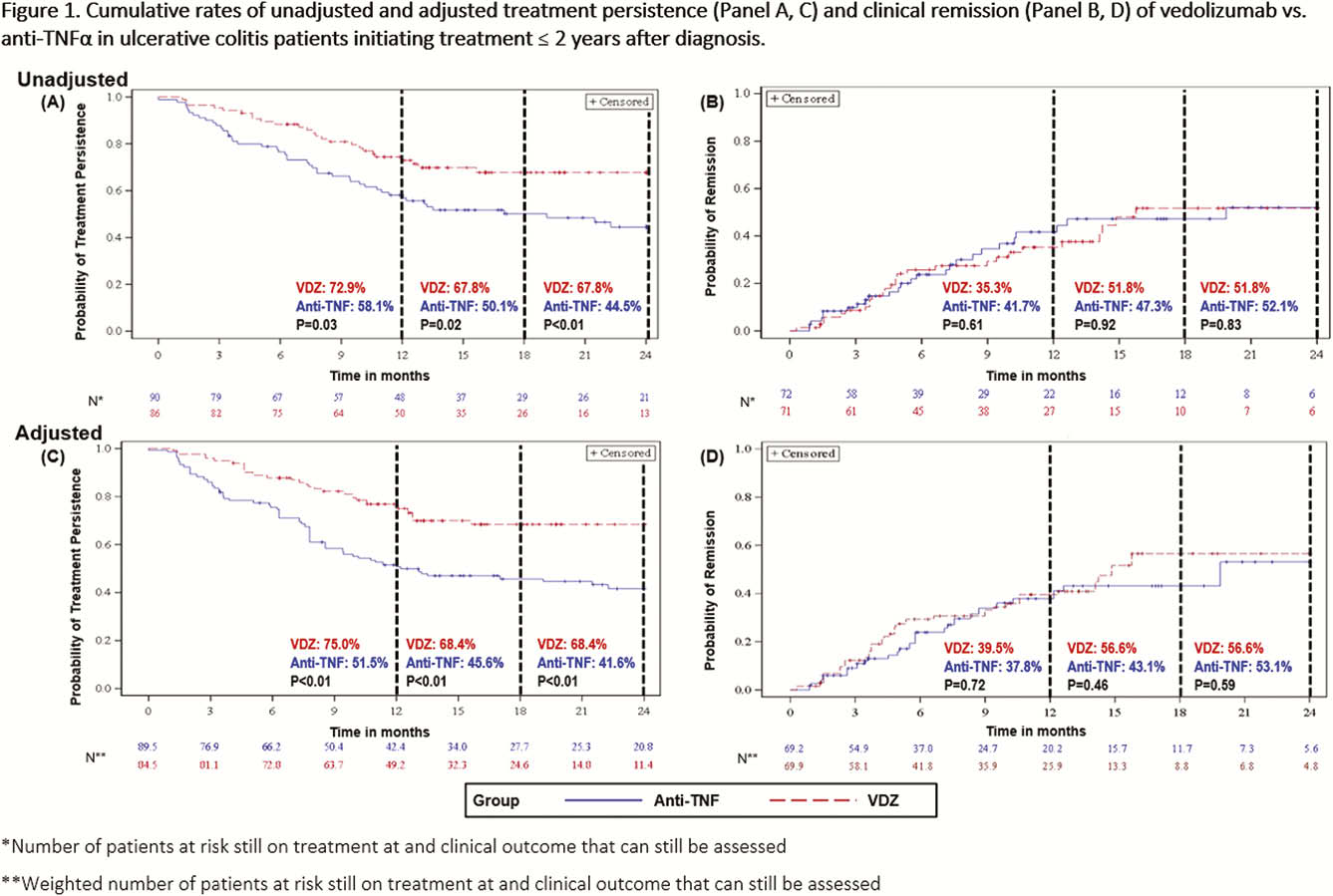
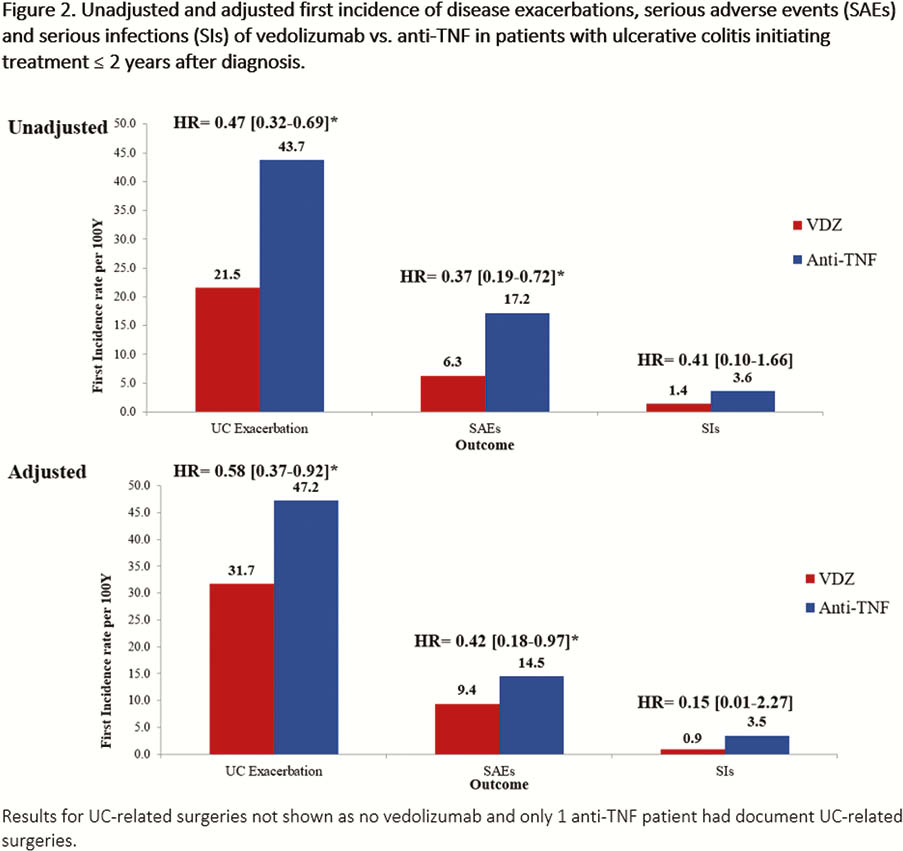
This was a real-world, multi-country, retrospective chart review study in Canada, Greece and the United States where biologic-naïve UC patients (≥18 years old) were treated with VDZ or an anti-TNF (adalimumab, infliximab, golimumab) agent within 2 years following diagnosis (initiated Tx May 2014–March 2018). Clinical effectiveness and safety data were collected from Tx initiation to earliest of chart abstraction date, death, or 6 months post-Tx discontinuation (Canada only). Tx persistence was defined as the duration of time from treatment initiation to discontinuation. Analyses of cumulative rates of Tx persistence, clinical response, clinical remission and mucosal healing over 24 months were estimated using Kaplan–Meier analyses. Clinical response, remission and mucosal healing were assessed using pre-defined hierarchical algorithms of standard disease measures reported in the medical records. Analyses of incidence rates (per 100 person-years [PYs]) of disease exacerbations, disease-related surgeries, serious adverse events (SAEs) and serious infections (SIs) were performed. Adjusted analyses used inverse probability weighting to balance cohorts.
Results
This analysis included 176 UC patients (VDZ: 86; anti-TNF: 90) from 37 sites. Mean (SD) age at index date: VDZ, 41.4 (18.9); anti-TNF, 36.8 (15.6) years (
Conclusion
EVOLVE is one of the first studies that compared early VDZ Tx to early anti-TNF Tx in biologic-naïve UC patients. Results showed VDZ was associated with higher persistence, lower likelihood of experiencing disease exacerbations and a more favourable safety profile. Thus, in early UC, Tx with VDZ may improve long-term clinical outcomes. Sample size limitations warrant further study.


