DOP59 Maintenance of remission with tofacitinib in patients with ulcerative colitis: Updated results of a subpopulation analysis from an open-label, long-term extension study, OCTAVE Open
J.F. Colombel1, M.T. Osterman2, P. Ibanez3, A.J. Thorpe4, H. Zhang4, N. Lawendy4, C. Su4, W. Reinisch5
1Icahn School of Medicine at Mount Sinai Hospital, New York, New York, USA, 2Penn Presbyterian Medical Center, Philadelphia, PA, USA, 3Clínica Las Condes, Santiago, Chile, 4Pfizer Inc., Collegeville, PA, USA, 5Medical University of Vienna, Vienna, Austria
Background
Tofacitinib is an oral, small-molecule JAK inhibitor for the treatment of ulcerative colitis (UC). Efficacy and safety were demonstrated in 3 Phase 3, randomised, placebo-controlled studies in patients with moderate to severe UC.1 An ongoing, Phase 3, open-label, long-term extension (OLE) study (OCTAVE Open, NCT01470612) included patients from OCTAVE Induction 1 and 2 and OCTAVE Sustain.
Methods
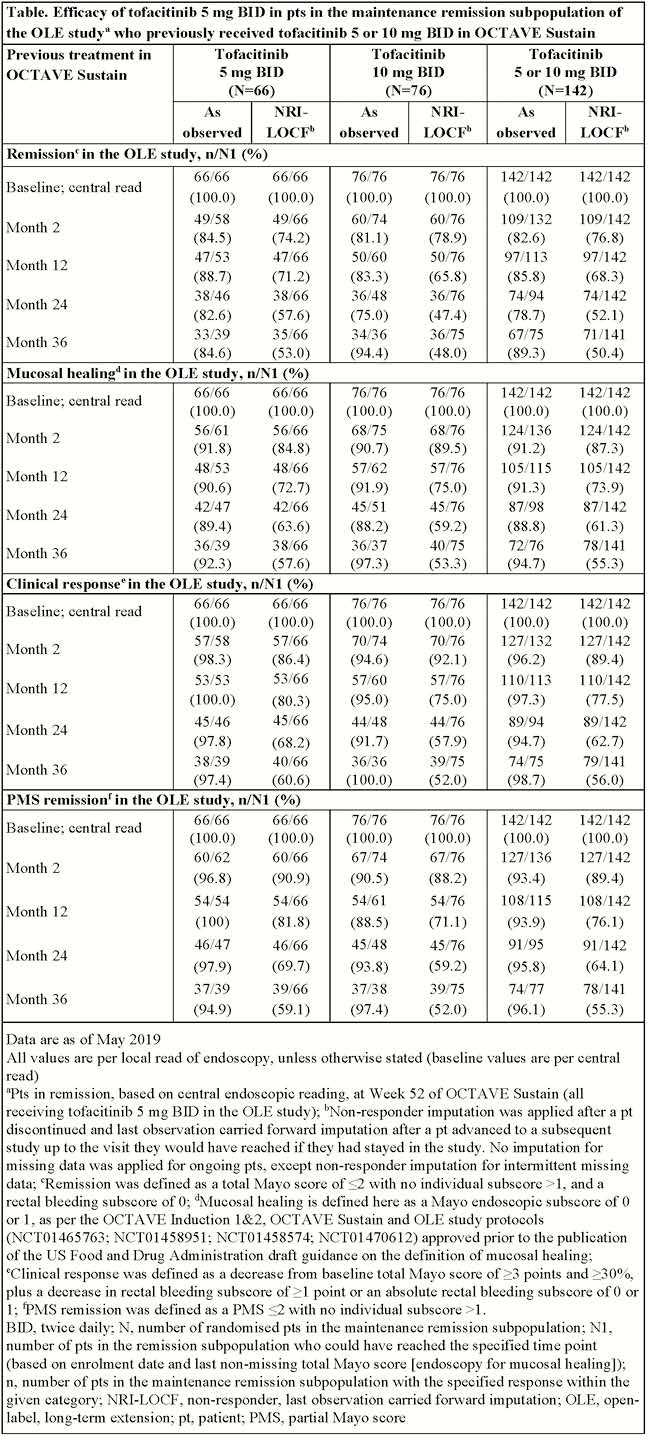
We present data (as of May 2019) from the ‘maintenance remission’ subpopulation in the OLE study who were in remission (total Mayo score ≤2, no individual subscore >1, rectal bleeding subscore of 0) at Week 52 of OCTAVE Sustain (having received tofacitinib 5 or 10 mg twice daily [BID]). These patients received tofacitinib 5 mg BID as per protocol in the OLE study. Efficacy data up to Month 36 of the OLE study (as observed and with non-responder and last observation carried forward imputation [NRI-LOCF]) are presented for this subpopulation. Safety data are reported for all patients who received tofacitinib 5 mg BID in the OLE study.
Results
Of 944 patients receiving ≥1 dose of tofacitinib in the OLE study, 163 were in remission at Week 52 of OCTAVE Sustain (mean age 45 years; 46.0% female). Of these, 66 (40.5%) and 76 (46.6%) received tofacitinib 5 and 10 mg BID, respectively, in OCTAVE Sustain, and 21 (12.9%) received a placebo. In total, 67/163 (41.1%) patients discontinued the OLE study, 16 (9.8%) due to adverse events (AEs) excl. worsening UC and 15 (9.2%) due to insufficient clinical response. Among patients that continued, efficacy (Table) was maintained over 36 months and was similar irrespective of the dose received in OCTAVE Sustain. Of 175 patients who received tofacitinib 5 mg BID (incl. 163 from the maintenance remission subpopulation), 152 (86.9%), 33 (18.9%) and 20 (11.4%) had AEs, serious AEs and severe AEs, respectively. The most frequent treatment-emergent AEs (TEAEs) were worsening UC (41 patients, 23.4%) and nasopharyngitis (38 patients, 21.7%). Six (3.4%) patients receiving tofacitinib 5 mg BID had serious infections, 11 (6.3%) had herpes zoster (non-serious and serious), 4 (2.3%) had opportunistic infections, 2 (1.1%) had major adverse cardiovascular events and 5 (2.9%) had malignancy (excl. non-melanoma skin cancer). No deep vein thrombosis, pulmonary embolism or deaths were reported in patients receiving tofacitinib 5 mg BID.
Conclusion
Most patients in remission at Week 52 of OCTAVE Sustain maintained efficacy with tofacitinib 5 mg BID over 36 months in the OLE study. Similar efficacy was observed for patients whose dose was reduced from tofacitinib 10 mg BID in OCTAVE Sustain to 5 mg BID in the OLE study, vs. those who received 5 mg BID throughout both OCTAVE Sustain and the OLE study. No new safety risks were identified.
Sandborn WJ


