DOP83 Intestinal fibrosis in Crohn’s disease patients is marked by up-regulation of innate immune cells and mucosal B cells
B. Creyns1,2, D. Gabriele3, C. Jonathan1,2, B. Verstockt2, G. Bislenghi4, F. Marc2,4, S. Vermeire2,4, G. Van Assche2,4, J. Ceuppens1, A. D’Hoore4, G. De Hertogh5, C. Breynaert1,6, Allergy and Clinical Immunology Research Group
1KU Leuven, Department of Microbiology, Immunology and Transplantation- Allergy and Clinical Immunology Research Group,Leuven, Belgium, 2KU Leuven, Department of Chronic Diseases, Metabolism and Ageing, Translational Research Center for Gastrointestinal Disorders TARGID, Leuven, Belgium, 3Clinical Gastroenterology Unit, Department of Biomedical Clinical and Experimental Sciences Mario Serio, University of Florence, Florence, Italy, 4Department of Gastroenterology and Hepatology, University Hospitals Leuven, KU Leuven, Leuven, Belgium, 5Department of Imaging and Pathology, Translational Cell and Tissue Research, KU Leuven, Leuven, Belgium, 6Department of General Internal Medicine, University Hospitals Leuven, KU Leuven, Leuven, Belgium
Background
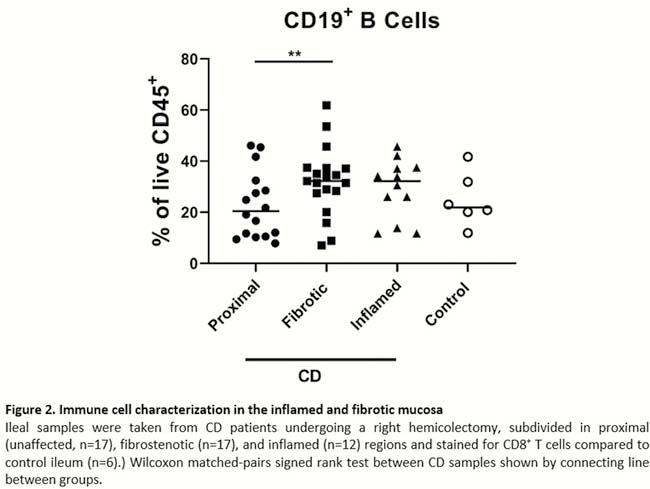
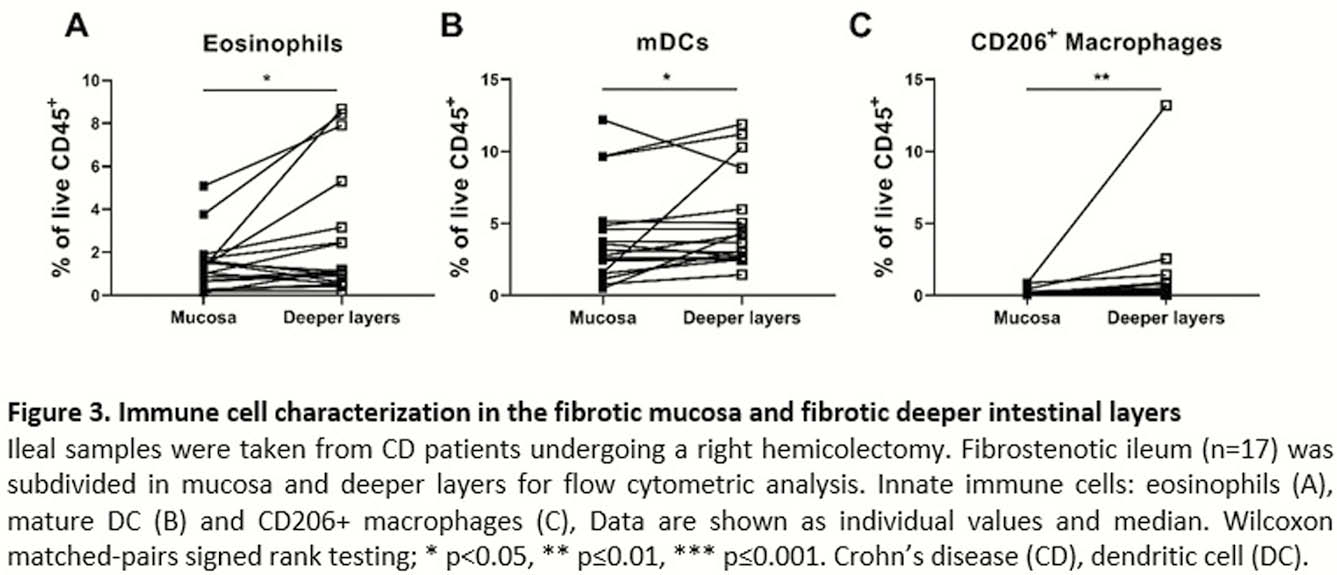
Intestinal fibrosis represents a significant complication of inflammatory bowel disease (IBD). An urgent need is present for the identification of pathways and markers involved in fibrogenesis to prevent and intervene with intestinal fibrosis. As intestinal fibrosis mainly occurs in the intestinal deeper layers, mucosal biopsies for studies in intestinal fibrosis are of limited information. Therefore, characterisation of the relative contribution of innate and adaptive immune cells in the mucosa and deeper layers in fibrotic distal ileum of Crohn’s disease (CD) patients was performed.
Methods
Seventeen CD patients undergoing right hemicolectomy (RHC) for stricturing disease and 6 colorectal cancer (CRC) patients undergoing RHC were recruited. The resected ileum was divided in macroscopically inflamed and fibrotic tissue and single-cell suspensions were made from mucosal and deeper intestinal layers for immune cell characterisation with flow cytometry. For comparison, proximal unaffected CD tissue and control ileum from CRC patients was included. Fibrosis and inflammation were confirmed on HE stained histological sections. Wilcoxon matched-pairs signed-rank test between CD samples and Kruskal–Wallis testing with Dunn’s multiple comparisons test compared with CRC ileum were performed.
Results
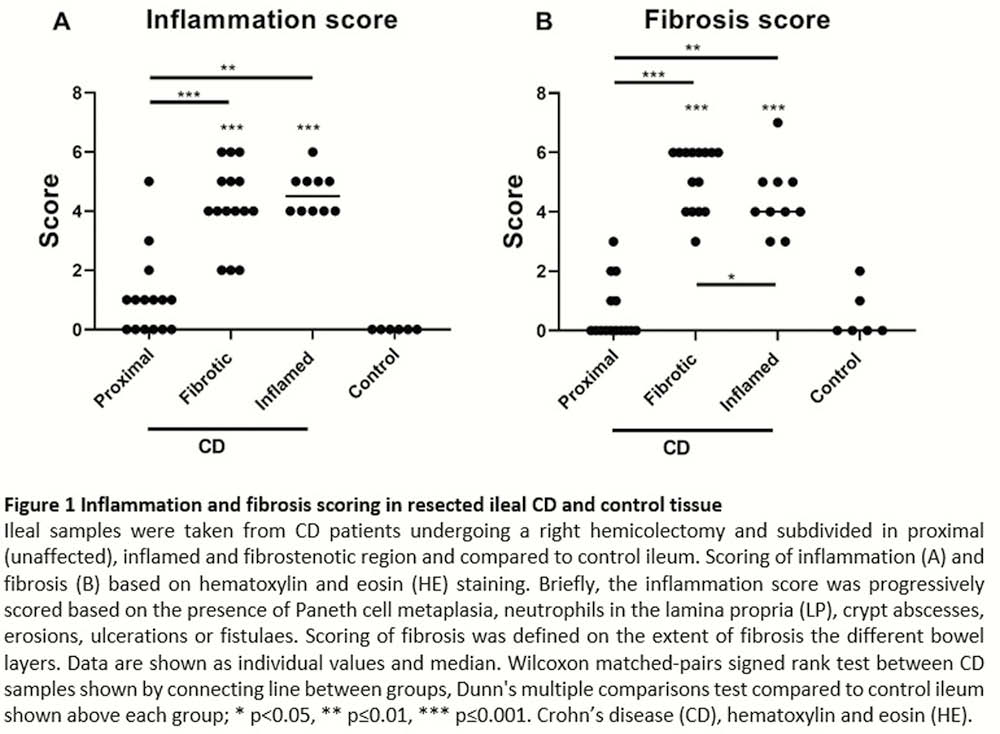
From 12 CD patients, an additional macroscopically inflamed region could be identified next to the fibrotic area, with a decreased fibrosis score as compared with the fibrotic area (4.00 vs. 6.00,
Conclusion
These results argue that inflammation in the deeper intestinal layers is different from the inflammatory signature seen in mucosal inflamed regions. We here report alternative innate immune cells expanded specifically in the deeper intestinal layers of fibrostenotic CD ileum that could identify targets for new anti-fibrotic therapies.


