OP06 The influence of different prednisolone tapering algorithms on the effectiveness of infliximab in patients with Ulcerative Colitis – A real-world cohort study
Ovesen, P.D.(1);Ilvemark, J.F.K.F.(1);Attauabi, M.(1);Seidelin, J.B.(1);
(1)Herlev Hospital- University of Copenhagen, Department of Gastroenterology and Hepatology, Herlev, Denmark;
Background
Data regarding the influence of prednisolone tapering on clinical outcomes among patients with ulcerative colitis (UC) are limited. We aimed to investigate the influence of different prednisolone tapering algorithms on the effectiveness of infliximab (IFX) among patients with UC.
Methods
This Danish retrospective single-center study included all patients with UC who were treated with IFX between 2009 and 2019 at Herlev University Hospital. The patients were grouped according to the prednisolone tapering: standard (≤5 mg/week), fast (>5 mg/week), or direct discontinuation after an initial course of less than one week. Finally, we included a control group of patients treated with IFX monotherapy. The primary outcome was corticosteroid-free clinical remission at weeks 14 and 52 defined as a partial Mayo score ≤1. Variables with a p-value ≤0.20 in univariable regression analysis were included in multivariable analysis. A subgroup analysis containing patients with acute severe ulcerative colitis (ASUC) treated with at least 40 mg of prednisolone at initiation of IFX was performed.
Results
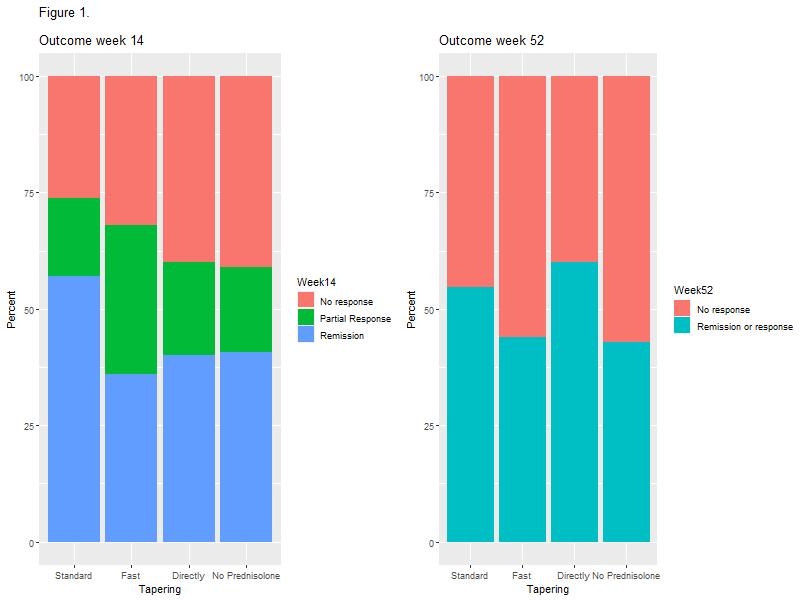
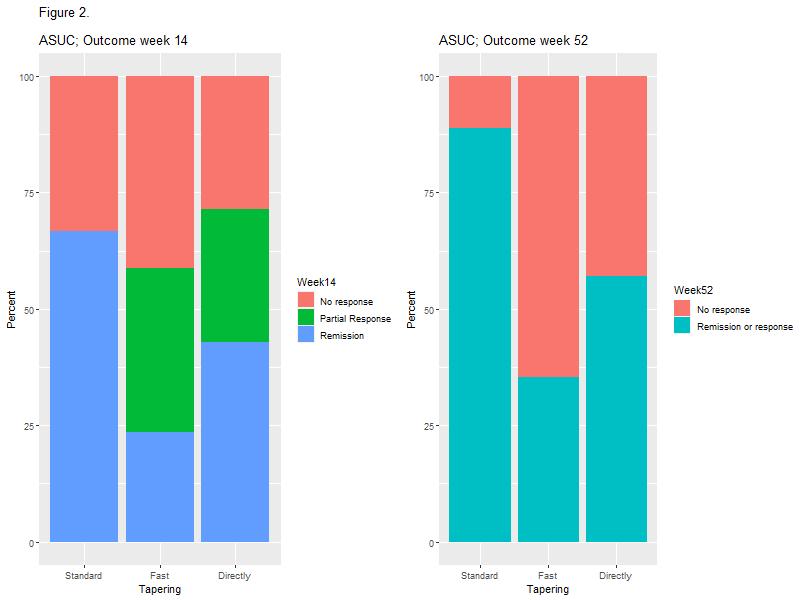
The study included 148 patients with UC of whom 81 (54.7%) were treated with prednisolone at the initiation of IFX. No association between prednisolone tapering and corticosteroid-free clinical remission with IFX at weeks 14 or 52 was observed (Figure 1 and Table 2). However, a higher proportion of patients in the standard tapering group achieved a C-reactive protein (CRP) level less than 5 mg/L at week 14 as compared with the fast-tapering group (23/23 (100%) vs. 14/18 (77.8%); p=0.03) and directly discontinuation group (6/10 (60%); p=0.03). This difference was not explained by prednisolone usage. In addition, none of the patients within the standard tapering regime (0/24; 0%) had severe activity at week 14 whereas this was seen in 4/19 (21.1%) in the fast tapering regime (p=0.03). In the subgroup analysis of 33 patients with ASUC, the standard tapering algorithm was associated with higher clinical remission as compared with the fast tapering regime at week 14 (9/14 (64.3%) vs. 5/19 (27.8%); p=0.02) and clinical response or remission at week 52 (12/14 (85.7%) vs. 7/19 (36.8%); p=0.01, Figure 2).
Conclusion
This study demonstrated no overall impact of prednisolone tapering algorithms on short and long-term effectiveness of IFX in patients with UC. However, standard tapering resulted in lower CRP levels and fewer cases of severe disease activity in the overall cohort and higher rate of short and long-term clinical response among ASUC patients, as compared with fast tapering regimes. Taken together, the data indicate that longer corticosteroid exposure in patients with high disease burden might improve IFX responses.


