P133 Point-of-care measurement of infliximab and adalimumab trough levels by the fluorescence resonance energy transfer (FRET) assay
Cerna, K.(1);Kastylova, K.(1);Sebek, P.(2);Luxova, S.(3);Lukas, M.(1);
(1)ISCARE a.s., Clinical & Research Center fo Inflamamtory Bowel Disease, Prague, Czech Republic;(2)Tanigen s.r.o., TaniGen, Prague, Czech Republic;(3)GENNET s.r.o., Immunological Laboratory, Prague, Czech Republic;
Background
Therapeutic drug monitoring (TDM) is the clinical practice of measuring serum drug concentrations to guide clinical decision-making. In IBD patients, it is applied mainly in infliximab (IFX) and adalimumab (ADL) therapy. TDM is either performed reactively in response to disease activity, or proactively in the absence of symptoms. The aim of the present study was to compare two analytical methods for the detection of trough infliximab and adalimumab levels (TL): point-of-care (POCT) fluorescence resonance energy transfer (FRET) method, and the enzyme- linked immunosorbent immunoassay (ELISA).
Methods
TL IFX were measured in n=24 and TL ADL in n= 21 blood samples. For ELISA, standard peripheral venous samples were used, and for POCT FRET measurements, capillary finger puncture was performed. Two assay results were compared evaluating the regression relationship using Spearman's correlation coefficient and the Bland-Altman plots. Cohen's kappa analysis was used to compare the degree of qualitative agreement between the two data sets. Repeatability was assessed by the difference of the repeated measurements, their means and standard deviations.
Results
The high degree of correlation was found between the measured values, both qualitative and quantitative (p < 0.0001 for both IFX and ADL), either expressed by the Spearman’s correlations, or by Bland-Altman analysis. Cohen’s weighted kappa 0.903 for IFX and 0.834 for ADL showed substantial/almost perfect agreement of qualitative results. Closeness of the agreement of successive measurements was very acceptable for both TLs of both drugs.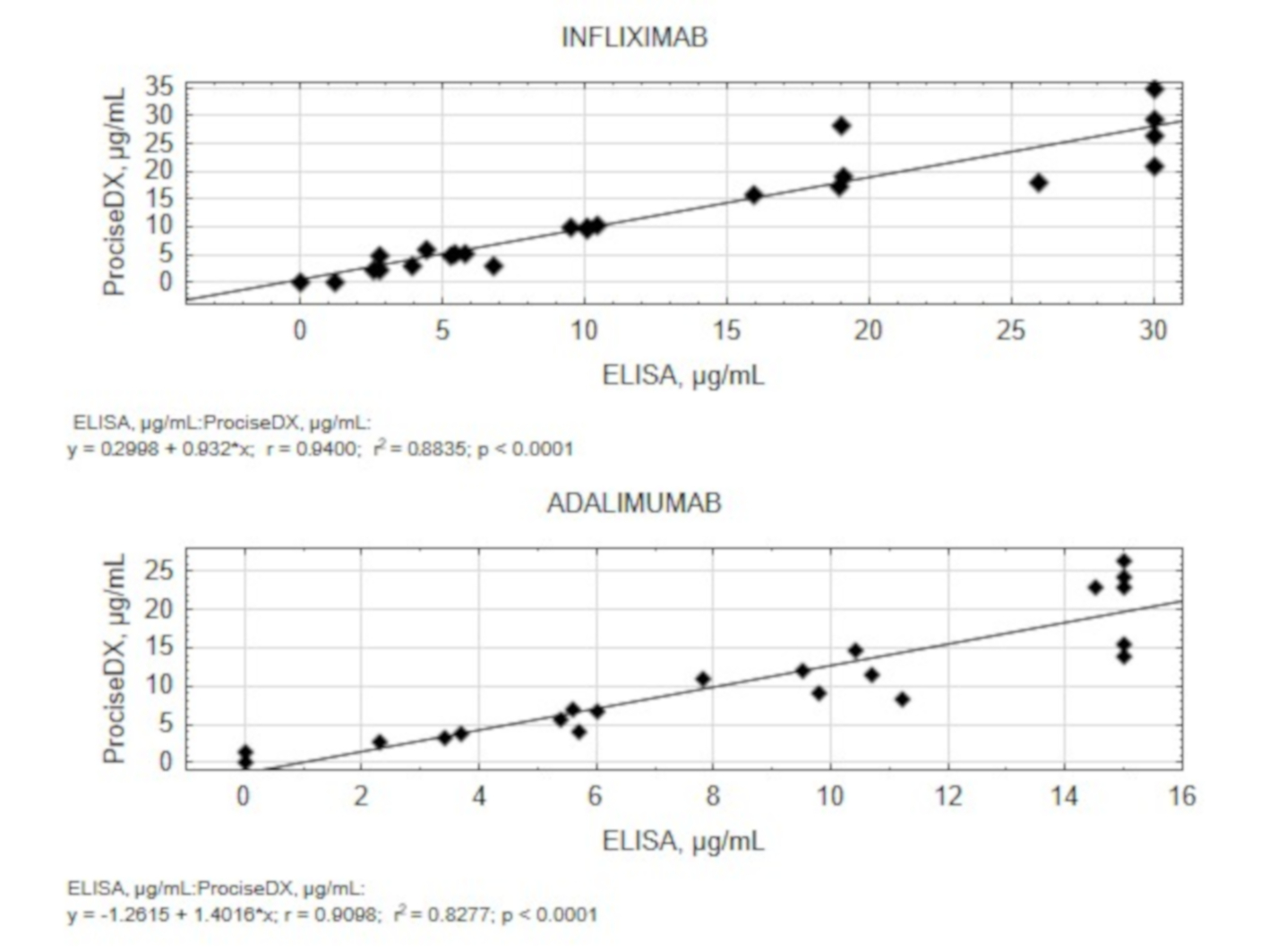
Conclusion
Point-of-care detection of IFX and ADL trough levels by FRET is accurate, comparable to the reference method, analysis is sufficiently consistent. For clinical sites with a proactive approach to TDM, FRET may be a useful tool for decision-making regarding individualization of biologic therapy.
Acknowledgements: We thank ProciseDX company for the ProciseDX analyzer rental, and for providing the laboratory diagnostics.


