P223 Degree of Histological Activity is not Associated with Vedolizumab Therapy Outcome in Ulcerative Colitis
Doherty, J.(1);Brennan, S.(2);Dinneen, K.(2);Muldoon, C.(2);Mc Kiernan, S.(1);Dunne, C.(1);Mac Carthy, F.(1);Hartery, K.(1);Ciara, R.(2);Kevans, D.(1);
(1)St James Hospital, Department of Gastroenterology, Dublin 8, Ireland;(2)St James Hospital, Department of Histopathology, Dublin 8, Ireland
Background
Histological inflammation is known to be associated with increased risk of disease relapse in patients with ulcerative colitis (UC). Vedolizumab (VDZ) is a gut selective anti-integrin which inhibits intestinal immune cell-trafficking. Whether the degree of histological activity at the time of VDZ therapy initiation is associated with therapy outcome is not known. We aimed to determine if there is an association between histological activity at the time of VDZ initiation and outcome of therapy.
Methods
A retrospective review was performed to identify UC patients treated with VDZ who had undergone an endoscopic assessment prior to therapy commencement. Baseline demographic data, information on therapy outcome and Mayo endoscopic sub score (MES) was collected for all patients. Endoscopic biopsies were retrieved and were scored for histological activity using the Geboes Score (GS). For Kaplan Meir analyses of primary endpoint, the cohort was dichotomised around a GS grade of 5. Primary endpoint was VDZ therapy outcome defined as persistence on VDZ therapy over time. Secondary endpoints included association between GS and MES and the association between a combined endoscopic and histological endpoint (MES = 3 & GS grade 5) and VDZ therapy outcome.
Results
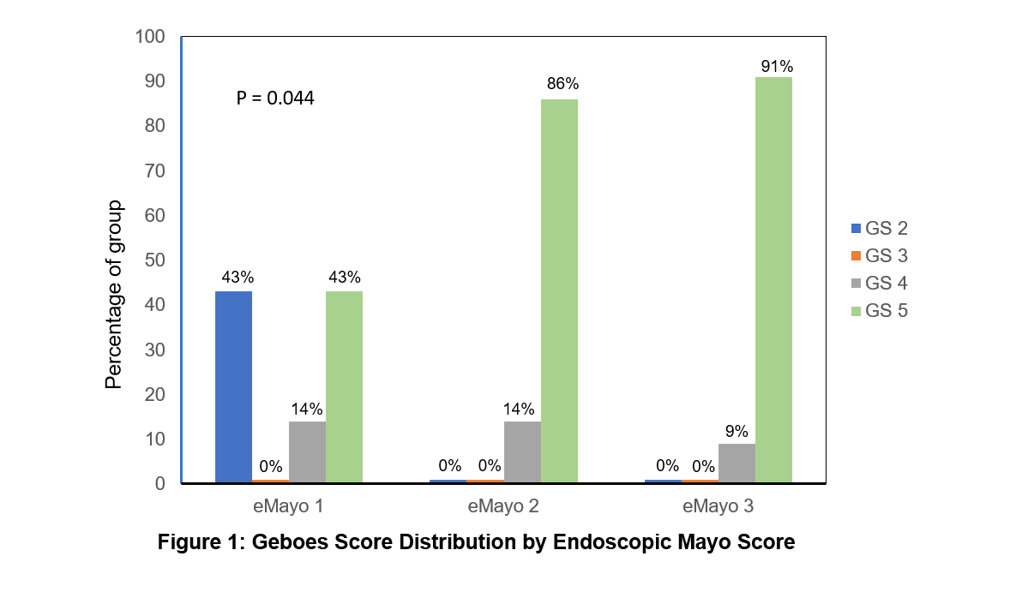
33 patients were included [median age 44.3 years (range 17.2 -84.3); 36% male gender]. 24%, 43% and 33% of the cohort had proctitis, left-sided colitis and extensive colitis respectively. 67% of subjects had prior anti-TNF exposure. Median time from endoscopy to commencement of VDZ was 9 weeks. Median study follow-up was 68 weeks (range 6.1 – 228.7). 3%, 21%, 42% and 33% had MES of 0, 1, 2 and 3 respectively. GS grade was significantly associated with MES (p = 0.04) (Figure 1). GS grade was not associated with time to discontinuation of VDZ (p=0.64) (Figure 2). Combined endoscopic and histological endpoint was not associated with time to discontinuation of VDZ (p=0.43) (Figure 3). The presence of lamina propria eosinophils was not associated with time to discontinuation of VDZ (p=0.92).


Conclusion
GS grade is associated significantly with MES which has been demonstrated previously. Neither histological activity alone nor in combination endoscopic activity were associated with outcome of VDZ therapy. Assessment of histological activity does not appear to provide additional information when selecting patients for VDZ therapy.


