P355 Acceptability of treatment regimen in inflammatory bowel disease: Results from a prospective nationwide study (ACCEPT2)
A. Buisson1, M. Fumery2, M. Serrero3, L. Orsat1, S. Nancey4, P. Rivière5, R. Altwegg6, L. Peyrin-Biroulet7, M. Nachury8, X. Hébuterne9, C. Gilletta10, M. Flamant11, S. Viennot12, G. Bouguen13, A. Amiot14, S. Mathieu15, L. Vuitton16, L. Plastaras17, A. Bourreille18, L. Caillo19, F. Goutorbe20, M. Fumery21, G. Pineton de Chambrun6, A. Attar22, X. Roblin23, B. Pereira24, B. Pariente8
1CHU Estaing, Department of Digestive and Hepatobiliary Medicine, Department of Gastroenterology, Clermont-Ferrand, France, 2CHU Amiens, Gastroenterology, Amiens, France, 3CHU Marseille, Gastroenterology, Marseille, France, 4CHU Lyon-Sud, Gastroenterology, Lyon, France, 5CHU Bordeaux, Gastroenterology, Bordeaux, France, 6CHU Montpellier, Gastroenterology, Montpellier, France, 7CHU Nancy, Gastroenterology, Nancy, France, 8CHU Lille, Gastroenterology, Lille, France, 9CHU Nice, Gastroenterology, Nice, France, 10CHU Toulouse, Gastroenterology, Toulouse, France, 11Private practice, Gastroenterology, Nantes, France, 12CHU Caen, Gastroenterology, Caen, France, 13CHU Rennes, Gastroenterology, Rennes, France, 14CHU Créteil, Gastroenterology, Créteil, France, 15Private practice- Pole Sante Republique ELSAN, Gastroenterology, Clermont-ferrand, France, 16CHU Besançon, Gastroenterology, Besançon, France, 17CH Colmar, Gastroenterology, Clermont-Ferrand, France, 18CHU Nantes, Gastroenterology, Nantes, France, 19CHU Nîmes, Gastroenterology, Nîmes, France, 20CH Bayonne, Gastroenterology, Bayonne, France, 21Private practice, Gastroenterology, Amiens, France, 22Private practice, Gastroenterology, Paris, France, 23CHU St Etienne, Gastroenterology, St-Etienne, France, 24CHU, Biostatistics unit, Clermont-Ferrand, France
Background
The multiplication of therapeutic options with close efficacy and safety, leads to consider acceptability of treatment regimen as a key point for therapeutic decision in inflammatory bowel disease (IBD). We aimed to compare acceptability of IBD treatment regimen and to identify its associated factors.
Methods
From a nationwide prospective study conducted in 24 public or private centres, IBD patients were consecutively included for 6 weeks. A dedicated questionnaire was developed, tested and validated for the study. It was administered to each included patient and his/her related physician. Acceptability was graded with an acceptability numerical scale (ANS) from 0 (absolutely not acceptable) to 10 (totally acceptable).
Results
Overall, 1850 patients were included (65.9% with Crohn’s disease (CD), mean age = 41.0 ± 14.7, 22.2% experiencing IBD flare at inclusion). The medications at inclusion were none in 9.0%, oral (PO) monotherapy in 17.3%, subcutaneous (SC) injections in 29.2% and intra-venous (IV) infusions in 44.5% of the patients. The ANS were 8.68 ± 2.52 for PO, 7.67 ± 2.94 for SC and 6.79 ± 3.31 for IV (
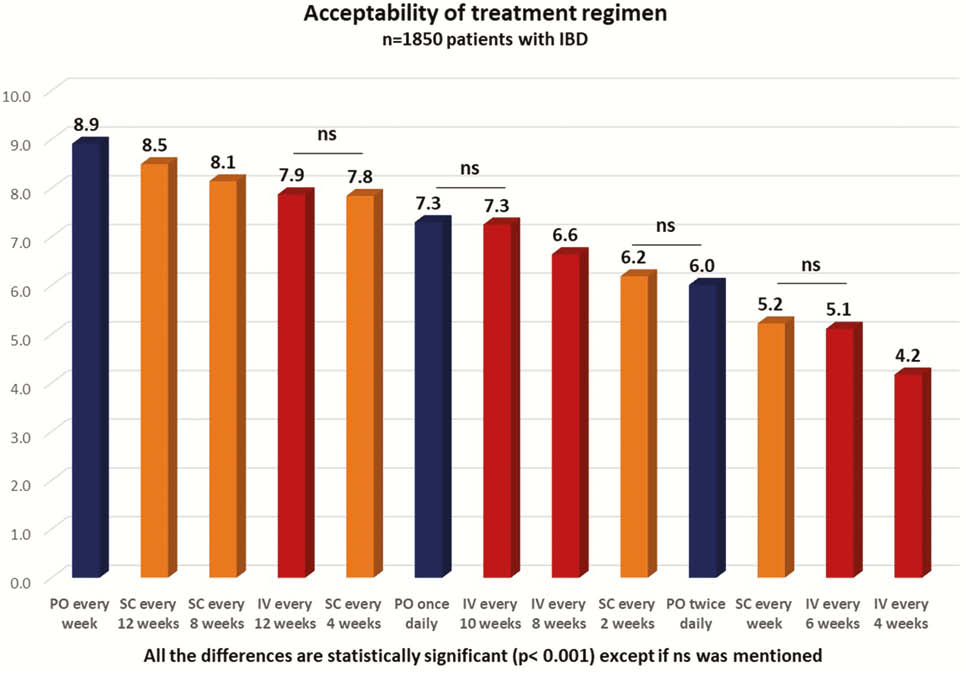
No difference was observed between CD and UC. In biologics-naïve patients (
Conclusion
While PO administration was preferred by most of IBD patients, the acceptability of treatment regimen is highly impacted by the interval between two doses and the previous medications. Our data could be helpful to guide therapeutic decision in daily practice in IBD.


