P389 Post induction infliximab trough levels predict long-term endoscopic remission in paediatric patients with inflammatory bowel disease
K. van Hoeve1, E. Dreesen2, I. Hoffman1, M. Ferrante3, S. Vermeire3
1Department of Paediatric Gastroenterology and Hepatology and Nutrition, University Hospitals Leuven, Leuven, Belgium, 2Laboratory for Therapeutic and Diagnostic Antibodies- Department of Pharmaceutical and Pharmacological Sciences, KU Leuven, Leuven, Belgium, 3Department of Gastroenterology and Hepatology, University Hospitals Leuven, Leuven, Belgium
Background
Although higher infliximab (IFX) trough levels (TL) have been associated with better outcomes, the ideal predictive sampling time and cut-points to achieve endoscopic remission remain unclear in children with inflammatory bowel disease (IBD). Therefore, we evaluated the pharmacokinetics of IFX during induction to predict long-term outcome of IFX.
Methods
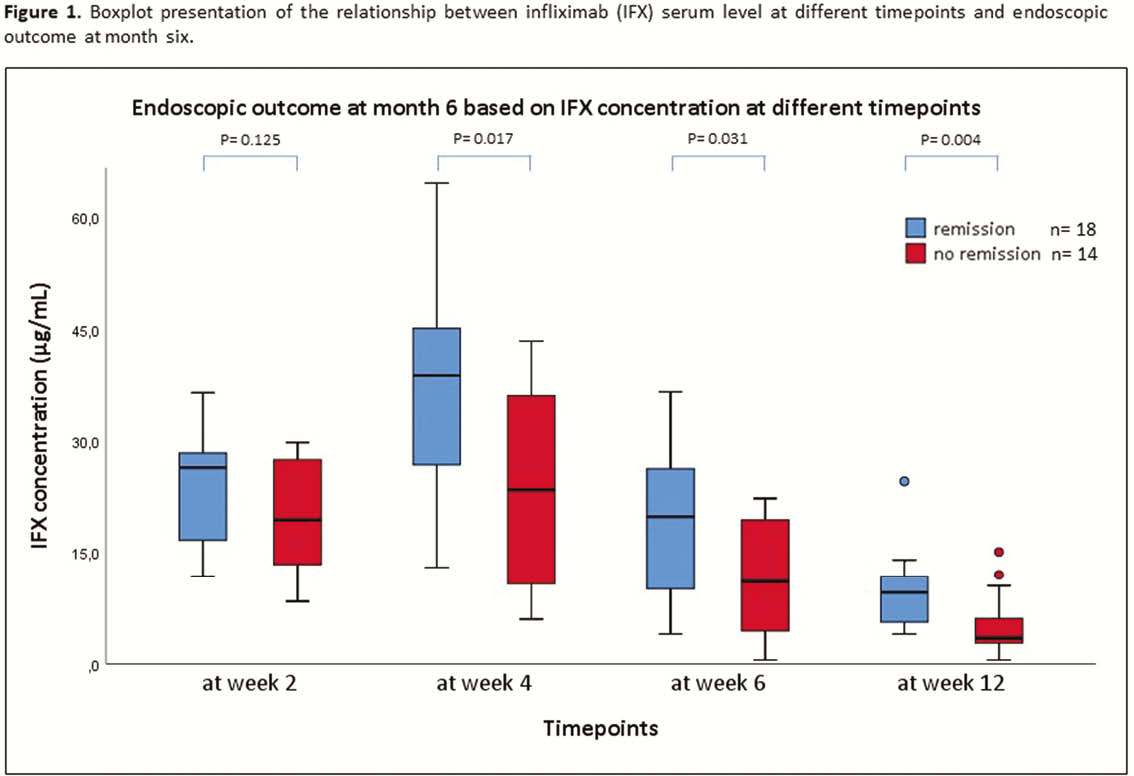
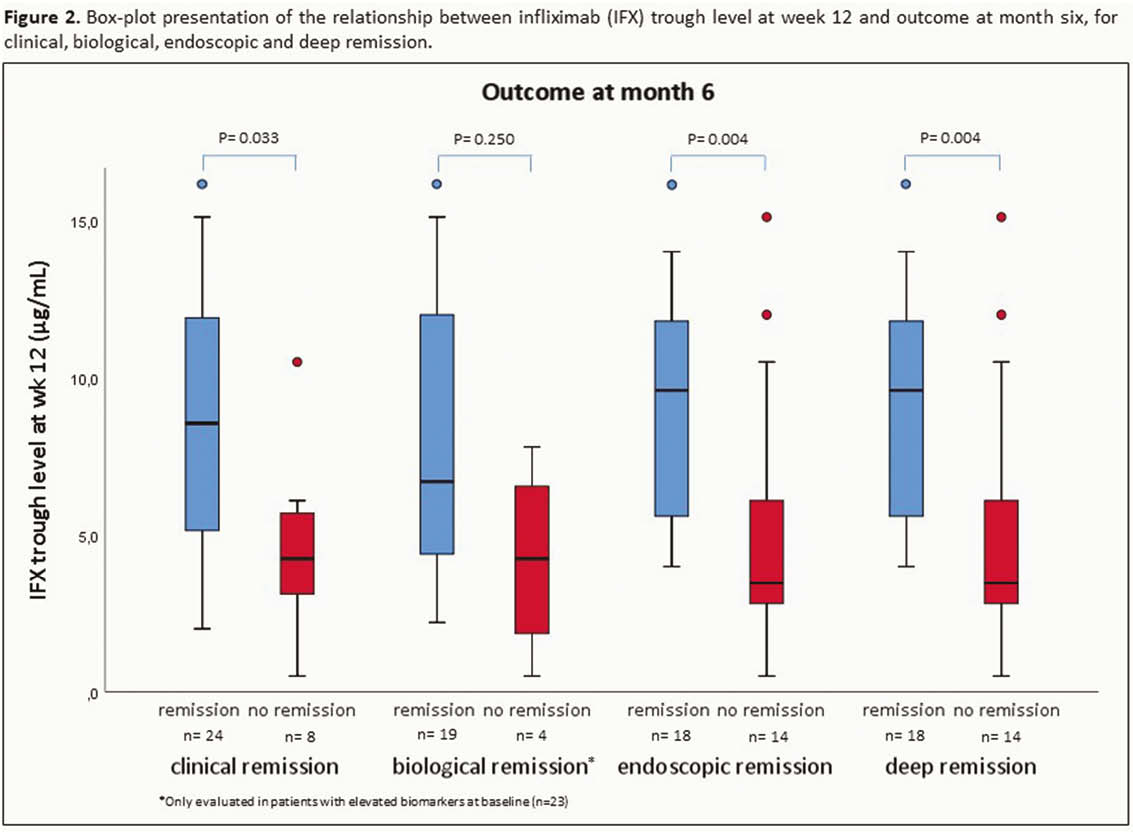
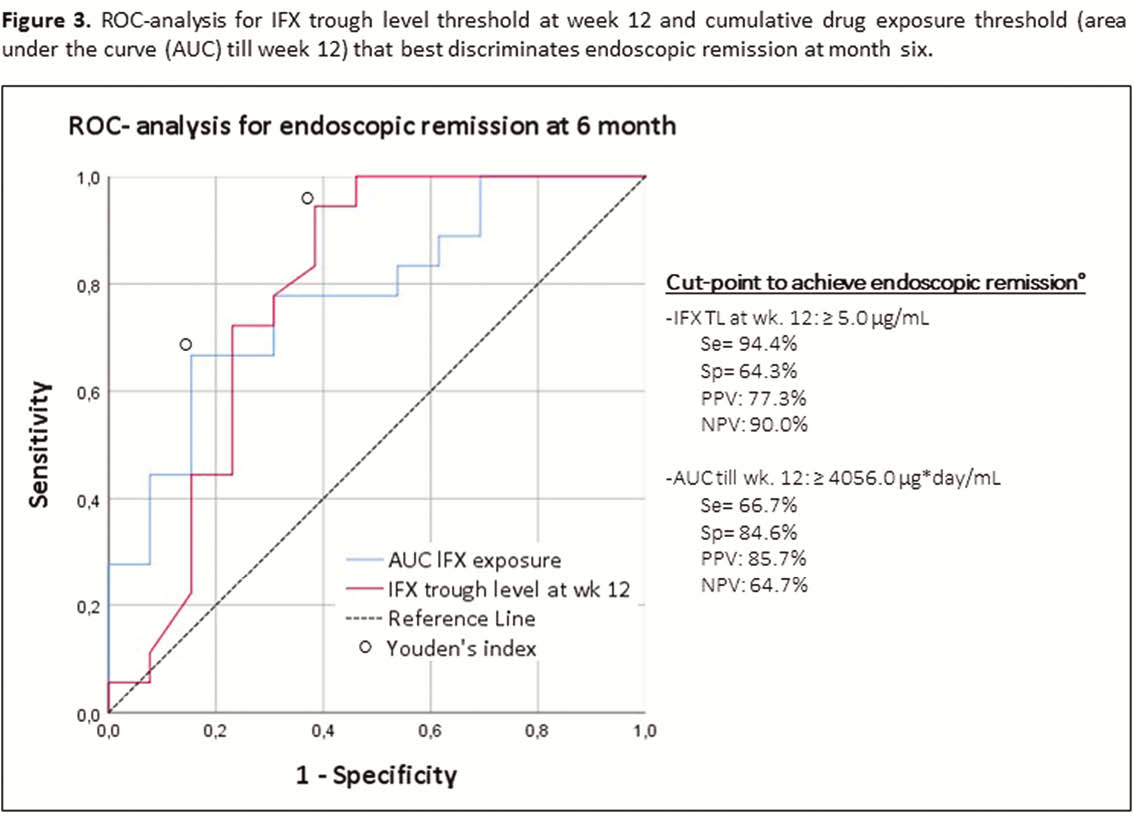
All children with Crohn’s disease (CD) or ulcerative colitis (UC) starting IFX therapy (5 mg/kg at weeks 0–2–6–12) for active luminal disease from May 2017 till May 2019 were followed prospectively. IFX levels were measured by Ridascreen IFX Monitoring ELISA (TL at weeks 2–6–12, peak at weeks 0–2–6 and intermediate at weeks 1–4). IFX levels and cumulative drug exposure (area under the curve (AUC) till week 12) were correlated with the outcome at month 6. Clinical remission was defined as PUCAI/PCDAI <10, biochemical remission as CRP ≤5 mg/l + ESR ≤20 mm/h, endoscopic remission as SES-CD <3 or Mayo endoscopic sub-score = 0 and deep remission if both clinical + endoscopic remission. Results were analysed using Mann–Whitney
Results
A total of 252 serum induction levels were included from 32 patients (20 CD and 12 UC; 38% male; median age at start of IFX 13.8 years [11.3–14.9]; 84% on concomitant thiopurines). Clinical remission was achieved in 24 (75%) patients and 18 (56%) were in endoscopic remission (all in deep remission) at month 6. Endoscopic remission at month 6 was associated with significantly higher median IFX TL at week 4 (38.8 µg/ml [24.3–46.0] vs. 23.5 µg/ml [10.5–36.6],
Conclusion
Adequate IFX exposure during induction in paediatric IBD patients is associated with significantly better clinical, endoscopic and deep remission rates at month 6. Model-informed precision dosing can assist physicians to achieve optimal exposure during induction more precisely (and rapidly) what is essential for an optimal outcome.


