P398 The early appearance of anti-drug antibodies during the induction phase predicts the clinical response of adalimumab and infliximab in IBD
X. Roblin1, N. williet1, P. veyrard1, G. boschetti2, A.E. berger3, S. nancey2, B. flourie2, S. paul3
1Department of Gastroenterology and Hepatology- Inserm- CIC1408, CHU Saint Etienne - Hospital Nord, Saint-Priest en Jarez, France, 2Department of Gastroenterology and Hepatology, CHU Lyon sud, Lyon, France, 3CHU Saint Etienne - Hospital Nord, Immunology, Saint-Priest en Jarez, France
Background
Several studies have shown a link between circulating anti-TNF levels during induction and clinical progression during IBD. There are less data on the detection of anti-drug antibodies at induction because it requires the use of so-called ‘drug-tolerant’ techniques. The purpose of our study was to investigate in two distinct cohorts (IBD patients on IFX or ADA) whether the early appearance of anti-drug antibodies measured using a drug-tolerant test was associated with clinical progression.
Methods
In a retrospective study, we included all patients followed in our department for at least 3 months and having had an anti-TNF dosage from W2 of induction under ADA or IFX. At follow-up, therapeutic failure was defined by the obligation to optimise, a change in biotherapy, the use of corticosteroids or surgery. IFX and ADA levels were measured routinely (Lisa-Tracker; Theradiag) and anti-medicine antibodies were detected by ‘drug-sensitive’ ELISA technique (Lisa-Tracker; Theradiag) or ‘drug-tolerant’ as previously described [1]. Patients with a positive rheumatoid factor were excluded.
Results
54 patients were included in ADA (mean age: 36 years, sex ratio M/F = 54%, CD: 65%, combotherapy: 10%) and 54 treated with IFX (mean age: 35 years, sex ratio M/F: 46%, CD: 62%, combotherapy: 65%). The search for anti-drug antibodies to W2 using a ‘drug-sensitive’ technique did not reveal any antibodies, whereas 74% of patients on ADA (AAA > or = 2.5 µg/ml eq) and 55% of patients on IFX (ATI > or = 4µg/ml eq). Under ADA, the duration of treatment until loss of response was significantly shorter in the presence of AAA at W2: 6 months vs. 24 months (HR = 18.51; 95CI = 4.35–78.11;
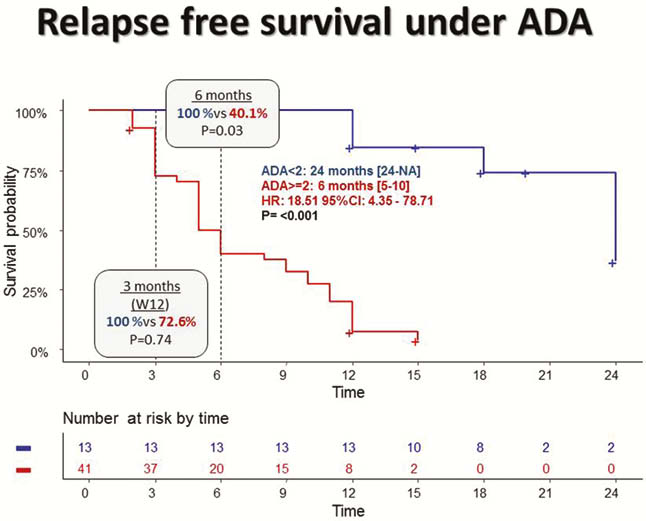
Conclusion
The early onset of anti-drug antibodies in the induction phase as early as W2 is the only isolated independent factor associated with loss of response. Anti-drug antibody frequencies are very high when measured with a drug-tolerant test and these Abs may be more neutralising with ADA than with IFX.


