P428 Oral mesalamine use in Crohn’s disease after implementation of the American College of Gastroenterology guidelines: a TARGET-IBD cohort study
N. Gupta1, B.E. Sands2, M. Regueiro3, M. Dubinsky4, D. Rubin5, J. Hanson6, F. Aberra7, M. Weiss8, S. Watkins9, D. Gazis10, M. Long11
1Atlanta Gastroenterology, Gastroenterology, Atlanta, USA, 2Icahn School of Medicine at Mount Sinai Medical Center, Dr Henry D. Janowitz Division of Gastroenterology, New York, USA, 3Cleveland Clinic, Gastroenterology- Hepatology and Nutrition, Cleveland, USA, 4Ichan School of Medicine- Mount Sinai Hospital, Pediatric GI and Nutrition, New York, USA, 5Gastroenterology, University of Chicago, Chicago, USA, 6Atrium Health, Gastroenterology and Hepatology, Charlotte, USA, 7Gastroenterology, University of Pennsylvania, Philadelphia, USA, 8Gastroenterology, Gastro Florida, Tampa, USA, 9TARGET PharmaSolutions, Scientific and Medical Affairs, Durham, USA, 10TARGET PharmaSolutions, Statistics, Durham, USA, 11Gastroenterology, University of North Carolina at Chapel Hill, Chapel Hill, USA
Background
American College of Gastroenterology (ACG) guidelines for the management of adults with Crohn’s disease (CD) in March 2018 state that oral mesalamine, ‘should not be used…to treat CD.’ 1 This abstract estimates the prevalence of any use of mesalamine among patients with CD before and after the implementation of the guideline. It also aims to describe current utilisation patterns of mesalamine in CD by disease phenotype, location, and concomitant use of biologic agents.
Methods
TARGET-IBD is a longitudinal cohort of IBD patients receiving usual care in the USA; this abstract includes CD patients enrolled between March 2017 and December 2018. Medication use was captured as any use in the follow-up period. Use of mesalamine was stratified into patients who were on mesalamine alone or patients on mesalamine and a biologic. The proportion of patients on mesalamine was estimated in time windows after the policy change to discern penetration into usual practice.
Results
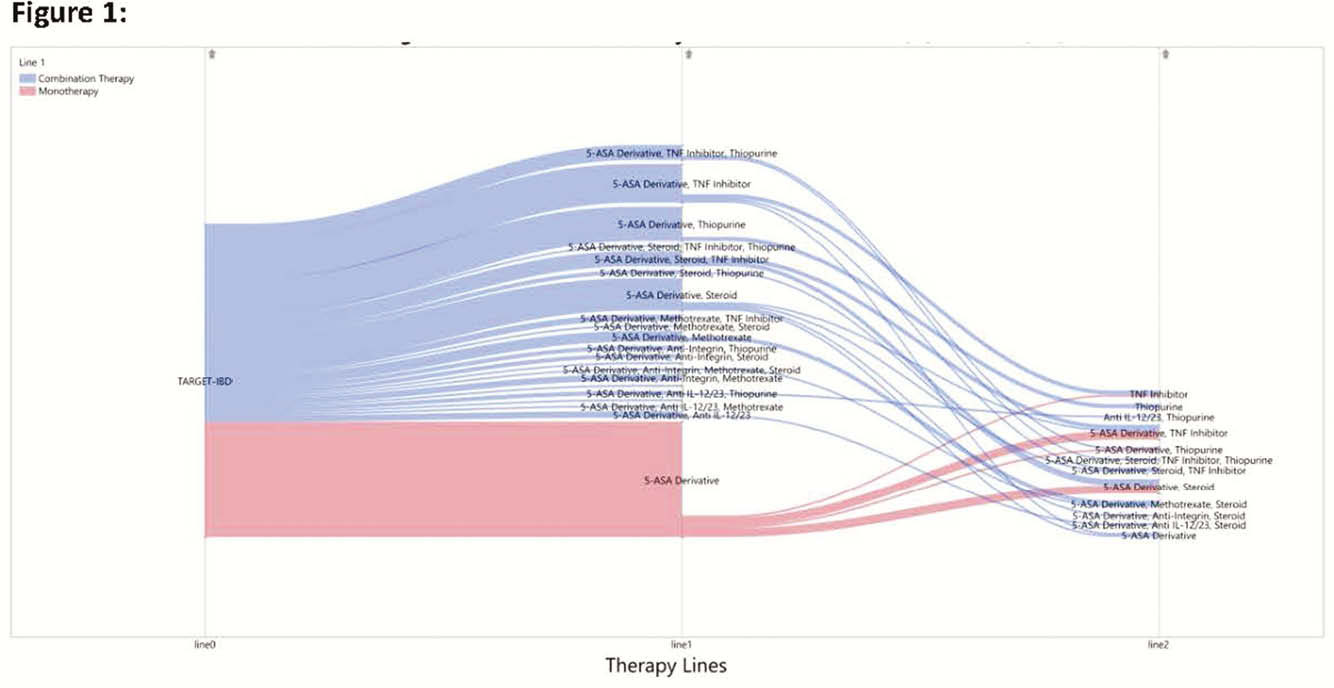
732 patients from TARGET-IBD who had baseline data prior to the new recommendation were included. Of these, 14% used mesalamine only, 8% used mesalamine plus a biologic, and 78% did not use mesalamine. A total of 65% received care in academic centres and the median duration of disease at enrolment was 12 years. The distribution of patient characteristics was similar after the policy change. Six months after the ACG recommendation, 11% of patients used mesalamine only, 7% of patients were on mesalamine plus a biologic, and 82% did not use mesalamine. Prevalence of use for the 3 and 6 month time windows was similar. Subsequent therapy changes and additions in patients on mesalamine are shown in Figure 1. Location (colon vs. ileum) and type of CD (inflammatory vs. fistulising) were statistically significant predictors of the odds of any mesalamine use both before and after the policy change (OR 3.233 (CI 1.725–6.061) and 3.334 (CI 1.931–5.755, respectively)).
Conclusion
Mesalamine use remained stable in the TARGET-IBD CD population, despite the release of the ACG guideline. Individuals with colonic, inflammatory disease were more likely to receive mesalamine, as were Medicare recipients. There was little difference between academic and community practice in implementation of the new standards. Given that the guidelines are relatively new, it is important to follow these participants over a longer period and monitor practice differences and ways to reinforce the practice recommendations to improve the quality and cost of care.


