P434 Effectiveness and safety of ustekinumab in ulcerative colitis: real-world evidence from Eneida Registry
M. Chaparro1, A. Garre1, M. Iborra2, M. Barreiro-de Acosta3, M.J. Casanova1, L. De Castro4, A. Fernández-Clotet5, J. Hinojosa6, M. Boscá-Watts7, D. Busquets8, A. López-García9, R. Lorente10, N. Manceñido11, T. Martínez12, D. Monfort13, J.L. Pérez-Calle14, A. Villoria15, T. Angueira16, F. Bermejo17, A.Y. Carbajo18, D. Carpio19, C. González-Muñoza20, F. Gomollón21, J.M. Huguet22, M. Rivero23, E. Iglesias-Flores24, M. Sierra25, L. Hernández-Villalba26, E. Domènech27, J.P. Gisbert1
1Hospital Universitario de La Princesa- IIS-IP- Universidad Autónoma de Madrid and CIBEREHD, Servicio de Aparato Digestivo, Madrid, Spain, 2Hospital Universitario y Politécnico La Fe and CIBEREHD, Gastroenterology Unit, Valencia, Spain, 3Complejo Hospitalario Universitario de Santiago, Gastroenterology Unit, Santiago de Compostela, Spain, 4Hospital Álvaro Cunqueiro-Complexo Hospitalario Universitario de Vigo, Gastroenterology Unit, Vigo, Spain, 5Hospital Clìnic, Gastroenterology Unit, Barcelona, Spain, 6Hospital de Manises, Gastroenterology Unit, Valencia, Spain, 7Hospital Clínico de Valencia. Universitat de València, Gastroenterology Unit, Valencia, Spain, 8Hospital Universitario de Girona Dr. Josep Trueta, Gastroenterology Unit, Gerona, Spain, 9Hospital del Mar and Institut Hospital del Mar d’Investigacions Mèdiques, Gastroenterology Unit, Barcelona, Spain, 10Hospital General Universitario de Ciudad Real, Gastroenterology Unit, Ciudad Real, Spain, 11Hospital Infanta Sofía, Gastroenterology Unit, San Sebastián de los Reyes, Spain, 12Hospital Virgen de la Luz, Gastroenterology Unit, Cuenca, Spain, 13Consorci Sanitari de Terrasa, Gastroenterology Unit, Barcelona, Spain, 14Hospital Universitario Fundación Alcorcón, Gastroenterology Unit, Madrid, Spain, 15Hospital de Sabadell. Depàrtament de Medicina. Universitat Autònoma de Barcelona y CIBEREHD, Gastroenterology Unit, Barcelona, Spain, 16Hospital General de Tomelloso and CIBEREHD, Gastroenterology Unit, Ciudad Real, Spain, 17Hospital Universitario Fuenlabrada and Instituto de Investigación Sanitaria del Hospital de La Paz IdiPAZ, Gastroenterology Unit, Madrid, Spain, 18Hospital Universitario Río Hortega, Gastroenterology Unit, Valladolid, Spain, 19Complexo Hospitalario Universitario de Pontevedra, Gastroenterology Unit, Pontevedra, Spain, 20Hospital de la Sta Creu i San Pau, Gastroenterology Unit, Barcelona, Spain, 21HCU Lozano BLesa- IIS Aragón and CIBEREHD, Gastroenterology Unit, Zaragoza, Spain, 22Hospital General Universitario de Valencia, Gastroenterology Unit, Valencia, Spain, 23Hospital Universitario Márques de Valdecilla and IDIVAL, Gastroenterology Unit, Santander, Spain, 24Hospital Universitario Reina Sofía, Gastroenterology Unit, Córdoba, Spain, 25Complejo Asistencial Universitario de León, Gastroenterology Unit, León, Spain, 26Hospital Santos Reyes, Gastroenterology Unit, Burgos, Spain, 27Hospital Universitario Germans Trials i Pujol and CIBEREHD, Gastroenterology Unit, Barcelona, Spain
Background
Ustekinumab has shown promising results in ulcerative colitis (UC) in the development program (UNIFI) that should be confirmed in clinical practice. Our aim was to evaluate the effectiveness and safety of ustekinumab in UC in real life.
Methods
Patients included in the prospectively maintained ENEIDA registry that received at least 1 dose of ustekinumab intravenously due to active UC were included. Clinical activity and effectiveness were defined based on Partial Mayo Score (PMS). The short-term response was assessed at week 8 and 16. The last-observation-carried-forward method was used in patients that stopped ustekinumab treatment before week 8 or 16. Variables associated with short-term remission were identified by logistic regression analysis. Data quality was assessed by remote monitoring.
Results
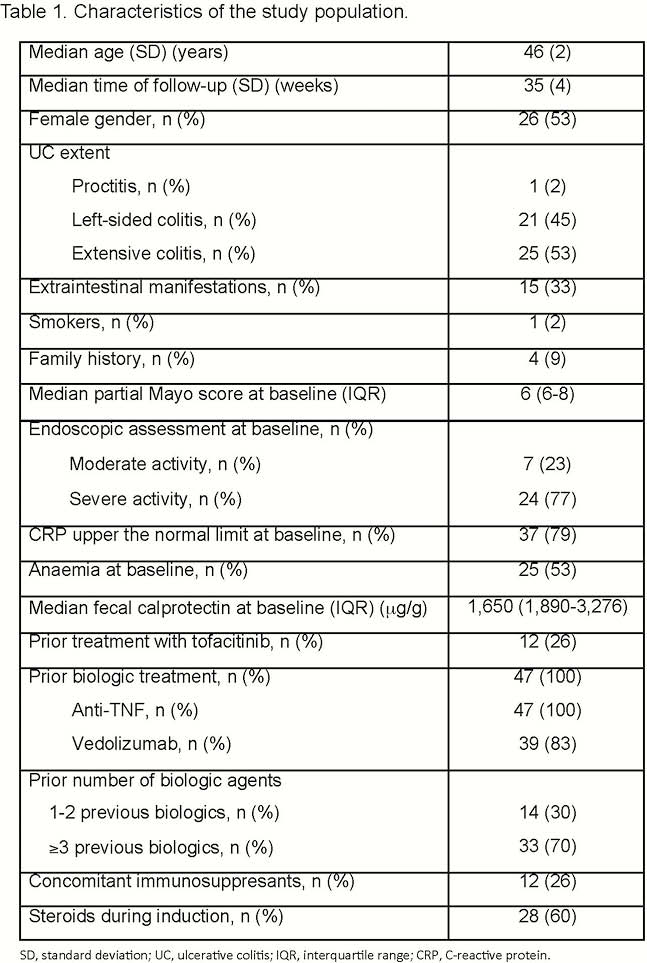
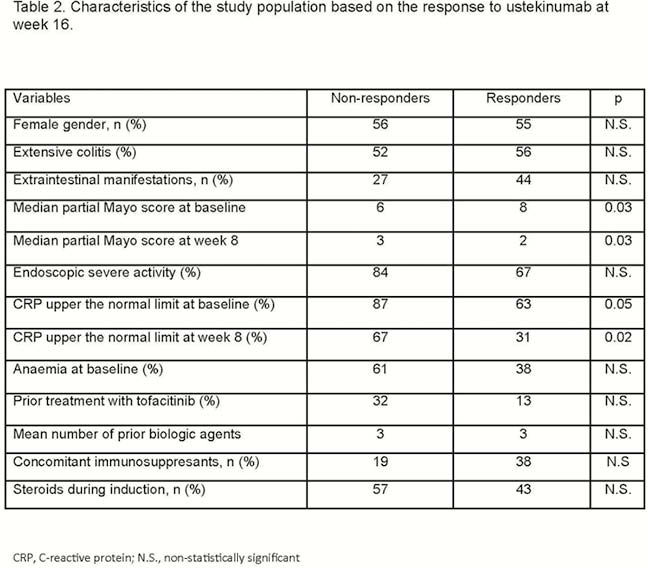
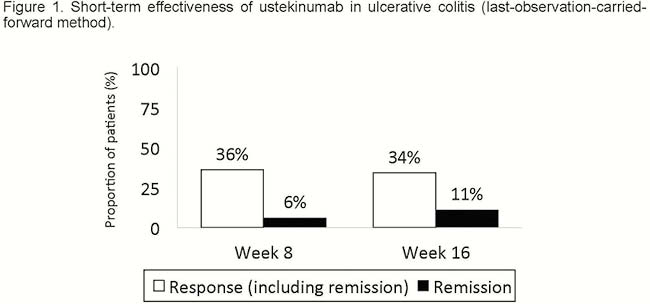
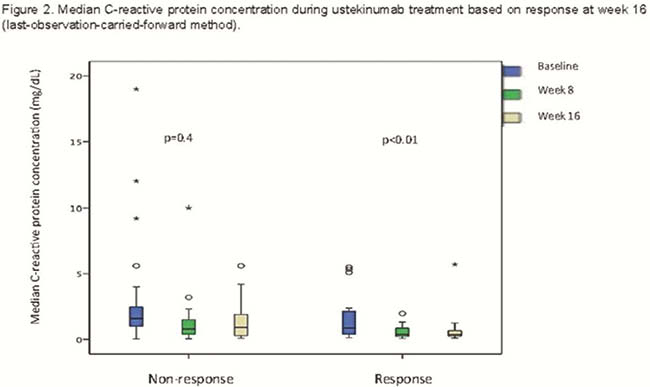
Forty-seven patients were included (Table 1); all of them had been previously exposed to biologic agents (70% to >2): 100% to anti-TNF and 83% to vedolizumab. A total of 26% had been exposed to tofacitinib. Seventeen patients (36%) had response at week 8 [3 of them (6%) had remission]; 16 patients (34%) had response at week 16 [5 of them (11%) had remission] (Figure 1). There was a statistically significant decrease in C-reactive protein (CRP) concentration during the induction only in patients with a response at week 16 (Figure 2). The proportion of patients with CRP elevated at baseline and at week 8 was higher among non-responders at week 16 (Table 2). In the multivariate analysis, higher PMS at week 8 [odds ratio (OR) = 0.5; 95% confidence interval (CI) = 0.3–0.9)] and CRP over the upper normal limit at week 8 (OR = 0.1; 95% CI = 0.01–0.8) were associated with lower probability of response at week 16; steroids during induction increased the probability of response at week 16 (OR = 8; 95% CI = 1–71). Of patients without response at week 8, only 7% achieved response at week 16. Seventeen out of 31 patients continued ustekinumab beyond week 16, despite being non-responders. Of these 17 patients, 4 reached remission after the third dose, 1 after the fifth and 1 after the seventh one. There were 2 infections, one of them with fatal consequences (in a patient under steroids and tacrolimus due to renal transplant).
Conclusion
Ustekinumab shows benefit in some UC patients in real practice, even in a very refractory cohort in which the drug was prescribed as last resort. Patient status at week 8 seems to be a good predictor of response after the induction. Safety was consistent with the known profile of ustekinumab.


