P511 Association of ustekinumab serum concentrations and clinical outcomes: results from the mUST-DECIDE trial
B. Bressler1, B. Sattin2, M.A. Williamson2, R. Khanna3, C.H. Seow4, D. Dajnowiec2, K. Karra2, L.L. Gao5, W. Afif6, mUST-Decide
1Department of Gastroenterology, University of British Columbia, Vancouver, Canada, 2Department of Medical Affairs, Janssen Canada, Toronto, Canada, 3Department of Epidemiology and Biostatistics, Western University, London, Canada, 4Department of Gastroenterology, University of Calgary Calgary, Canada, 5Research and Development- LLC, Janssen Inc., Spring House, USA, 6Inflammatory Bowel Disease Centre, McGill University Health Centre, Montreal, Canada
Background
Therapeutic drug monitoring is an important adjunct in optimising IBD management in patients treated with anti-TNF therapies. However, it remains unclear whether these principles apply to non-anti-TNF biologic therapies, such as the anti-IL12/23 agent, ustekinumab (UST). A follow-up analysis of the ‘mUST-DECIDE’ study explores the correlation between UST drug level and clinical outcomes.
Methods
111 consecutive UST-treated adult CD patients across 11 sites in Canada from April 2017 to January 2018 were evaluated. Clinical decisions were recorded for all subjects at the single study visit and blood was drawn for TDM (Sanquin, radioimmunoassay). A retrospective chart review was completed in patients who had a follow-up visit ≥ 30 days after the baseline visit. Improved disease control (primary outcome) was defined as a composite assessment outcome meeting ≥ 1 disease control criterion (symptomatic, endoscopic/imaging, biochemical) without any of the non-response criteria (inadequate or loss of response, worsening of any disease control criteria, initiation of any CD-related medications and adverse events). Serum [UST] were categorised as therapeutic, subtherapeutic or uninterpretable based on their position in a 2 compartment PK model [subjects on Q8W dosing were assessed as per the log-linear model which projected a therapeutic level of ≥4.5 µg/ml at 4 weeks and ≥1.0 µg/ml at 8 weeks].
Results
53 patients had a clinical follow-up visit and an interpretable serum [UST]. Patients had refractory disease (89% anti-TNF-exposed, median 16.4 years since diagnosis), but clinically well (66% had baseline HBI <5) on treatment with UST for a median [IQR] of 16.5 [10.4–26.8] months. At baseline, the majority had serum [UST] in the therapeutic range (
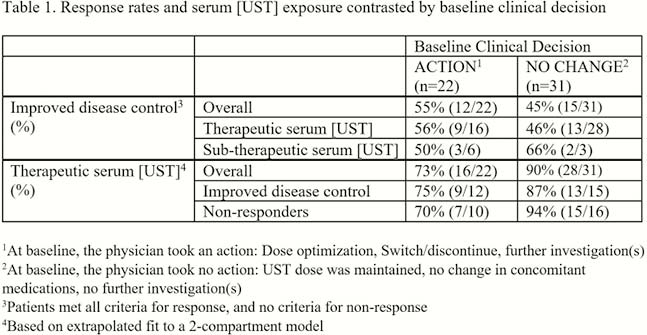
Conclusion
The sub-study did not detect any association between routine serum [UST] and short-term clinical outcomes at the next patient visit. These data broadly suggests that the routine measurement of serum UST in CD patients does not aid in disease management, though further studies across different clinical scenarios (e.g. loss of response) are warranted.


