P525 Rheumatoid arthritis treatment with filgotinib: Week 156 safety and efficacy data from a phase 2b open-label extension study
R. Besuyen1, A. Kavanaugh2, R. Westhovens3, K. Winthrop4, S. Lee5, J. Greer6, A. DeZure6, D. An7, L. Ye7, J. Sundy6, L. Meuleners8, R. Alten9, M. Genovese10
1Galapagos NV, Clinical Development, Mechelen, Belgium, 2Department of Rheumatology, University of California, San Diego, CA, USA, 3Department of Rheumatology, University UZ Leuven, Leuven, Belgium, 4Department of Rheumatology, Oregon Health and Science University, Portland, OR, USA, 5Gilead Sciences Inc., Department of Medical Affairs, Foster City, CA, USA, 6Department of Clinical Research, Gilead Sciences Inc., Foster City, CA, USA, 7Department of Biostatistics, Gilead Sciences Inc., Foster City, CA, USA, 8Department of Biostatistics, Galapagos NV, Mechelen, Belgium, 9Department of Internal Medicine and Rheumatology, Schlosspark-Klinik, Berlin, Germany, 10Division of Immunology and Rheumatology, Stanford University, Palo Alto, CA, USA
Background
Filgotinib (FIL) is an oral, selective Janus kinase inhibitor, shown to be effective and well tolerated in patients (pt) with rheumatoid arthritis (RA) and other inflammatory diseases (IBD/AS/PsA). DARWIN 3, an ongoing, open-label, long-term extension of phase 2b studies evaluates the longer-term safety and efficacy of FIL in RA.
Methods
The study evaluated pt outcomes for methotrexate (MTX) inadequate responders completing the 24-week DARWIN 1 (FIL+MTX) and DARWIN 2 (FIL monotherapy) studies. We present an interim analysis at week 156 following FIL 200 or 100 mg/day treatment. Event rate: total events/total years of exposure of FIL; Exposure: until data cut-off in patients on the study at the time of analysis.
Results
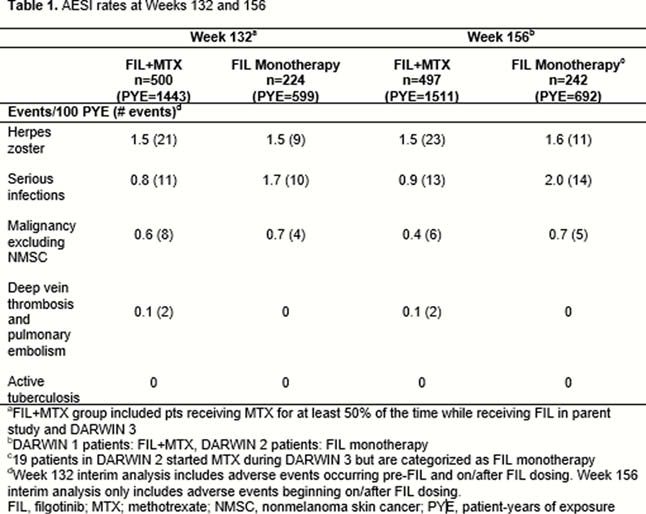
Eight hundred and seventy-seven patients completed the parent studies. Seven hundred and thirty-nine enrolled in DARWIN 3 (497 DARWIN 1, 242 DARWIN 2); most DARWIN 1 and 2 patients were female (81.5%, 81.8%), white (75.3%, 74.8%); mean age was 53 and 52 years, respectively. Mean baseline MTX dose in the FIL+MTX group: 16.8mg/week. At week 156, 59.9% of patients remained on study. Most common reasons for discontinuation were adverse events (26.5%) and subject request (9.1%). Total exposure to FIL was 2203 pt-years; mean exposure ± standard deviation (SD): 3.04 ± 1.22 years for FIL+MTX and 2.86 ± 1.21 years for FIL monotherapy. Treatment-emergent adverse events (TEAEs): 419 (84.3%) patients on FIL+MTX and 203 (83.9%) patients on FIL monotherapy; serious TEAEs occurred in 45 (9.1%) and 33 (13.6%), respectively. Adverse event of special interest (AESI) rates remained low at week 156 (Table 1). Grade ≥3 toxicities in >1% of patients were decreased lymphocytes (4.2%/1%), decreased neutrophils (1.0/1.2%), increased fasting triglycerides (2.2/0%) and decreased fasting triglycerides (0/3.3%) for FIL+MTX/FIL monotherapy respectively. 5 deaths occurred (FIL+MTX: 2, FIL monotherapy: 3) none after week 132. Clinical efficacy was shown at week 156 in both FIL+MTX and FIL monotherapy groups, using observed cases as measured by AC
Conclusion
FIL was generally well tolerated; no new safety signals emerged. No safety differences observed in patients receiving FIL+MTX or FIL monotherapy. Efficacy was sustained up to week 156 in both treatment groups.


