P572 Clinical outcomes after discontinuation of anti-tumour necrosis factor-α agents in inflammatory bowel disease patients with clinical remission: KASID multicenter study
J.H. Song1, S.M. Kong1, J. Shin1, G. Seong1, E.A. Kang2, S.K. Park3, S.N. Hong1, Y.S. Kim4, K.B. Bang5, K.O. Kim6, H.S. Lee7, S.B. Kang8, S.Y. Shin9, E.M. Song10, C.H. Choi9, T.J. Kim1, E.R. Kim1, D.K. Chang1, Y.H. Kim1
1Samsung Medical Center, Medicine, Seoul, Korea Republic of, 2Seoul National University College of Medicine, Internal Medicine and Liver Research Institute, Seoul, Korea Republic of, 3Kangbuk Samsung Hospital, Medicine, Seoul, Korea Republic of, 4Seoul Paik Hospital, Internal Medicine, Seoul, Korea Republic of, 5Dankook University College of Medicine, Internal Medicine, Cheonan, Korea Republic of, 6Yeungnam University College of Medicine, Internal Medicine, Daegu, Korea Republic of, 7Inje University Busan Paik Hospital, Internal Medicine, Busan, Korea Republic of, 8Daejeon St. Mary’s Hospital, Internal Medicine, Daejeon, Korea Republic of, 9Chung-Ang University, Internal Medicine, Seoul, Korea Republic of, 10Ewha Womans University Seoul Hospital, Internal Medicine, Seoul, Korea Republic of
Background
Despite proven efficacy of anti-tumour necrosis factor-α agents (ant-TNF) for inflammatory bowel disease (IBD), some patients have to be discontinuation of anti-TNF for various reasons in a real-world clinical setting. The aim of this study was to evaluate the long-term outcomes and risk factors of relapse after discontinuation of anti-TNF in IBD patients with clinical remission.
Methods
A retrospective multicenter cohort study was conducted at 10 referral hospitals, affiliated in IBD Study Group of the Korean Association for the Study of Intestinal Diseases. The study population comprised patients diagnosed with Crohn’s disease (CD) or Ulcerative colitis (UC) who had been treated with anti-TNF (infliximab (IFX) or adalimumab (ADA)) to induce remission and in whom ant-TNF had been discontinued after clinical remission was achieved. The patients were excluded for follow-up of <12 months after discontinuation of anti-TNF.
Results
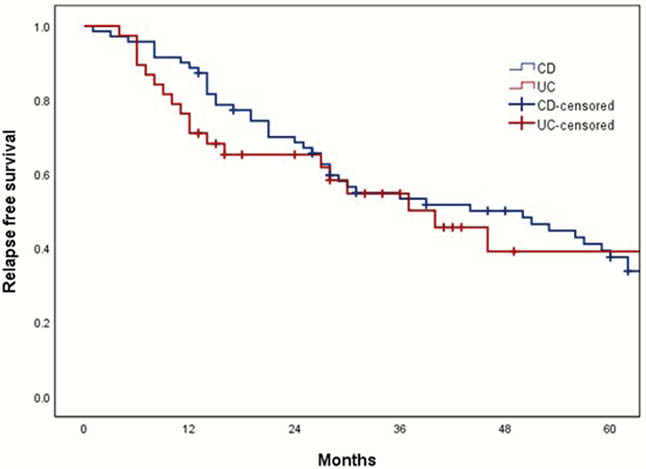
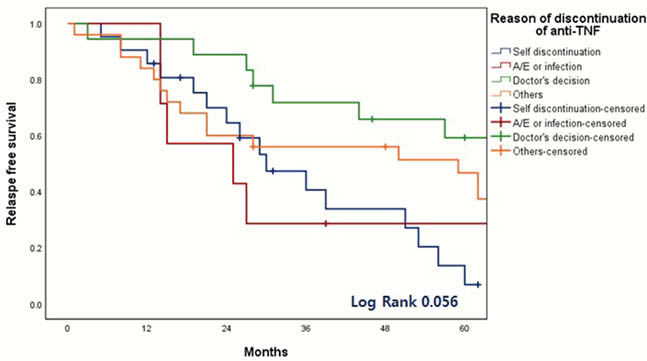
A total of 125 IBD patients were eligible. Among them, 109 IBD patients including 71 CD and 38 UC were analyzed and median follow-up period was 56 months (interquartile range, 35–90 months). The reasons of discontinuation of anti-TNF was physician’s decision (
Conclusion
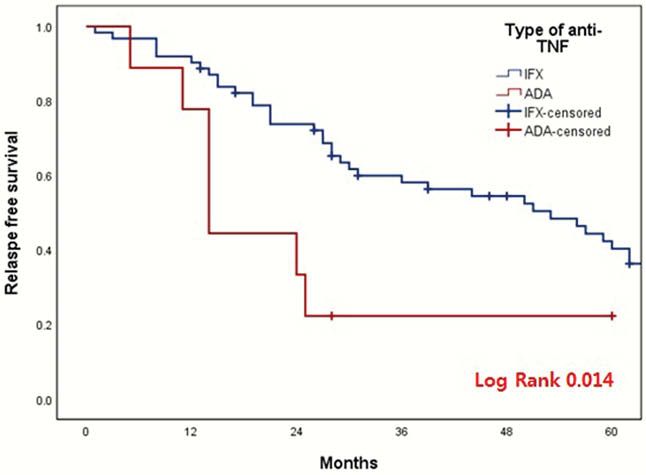
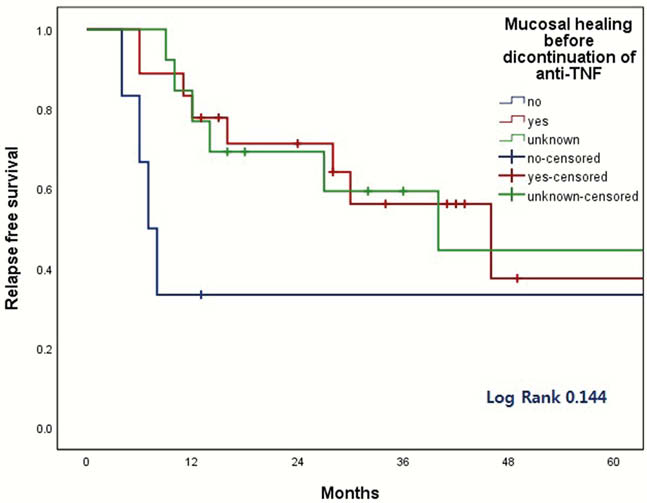
The discontinuation of anti-TNF was associated with increased risk of relapse. Although retreatment of anti-TNF seems to be effective and safe, the discontinuation of anti-TNF should be carefully considered based on the type of anti-TNF, the reason for discontinuation, and the mucosal healing status.


