P574 Incidence and indicators of suboptimal response to tumour necrosis factor antagonist therapy in Chinese patients with inflammatory bowel disease: Results from the EXPLORE study
J. Li1, Z. Li2, P. Hu3, Z. Wen4, Q. Cao5, X. Zou6, Y. Chen7, Y. Wang8, J. Zhong9, X. Shen10, D. Demuth11, O. Fedeeva11, L. Xie12, J. Chen12, J. Qian1
1Peking Union Medical College Hospital, Chinese Academy of Medical Sciences, Beijing, China, 2Shanghai Tenth People’s Hospital, Tongji University, Shanghai, China, 3The Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, China, 4West China Hospital, Sichuan University, Chengdu, China, 5Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou, China, 6Nanjing Drum Tower Hospital, Nanjing University School of Medical, Nanjing, China, 7The Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China, 8The First Affiliated Hospital, Dalian Medical University, Dalian, China, 9Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China, 10Zhongshan Hospital, Fudan University, Shanghai, China, 11Medical Affairs, Takeda Pharmaceutical International AG, Singapore, Singapore, 12Medical Affairs, Takeda China International Trading Company, Beijing, China
Background
Inflammatory bowel disease (IBD) patients may experience a suboptimal response to anti-tumour necrosis factor (TNF) therapy. In China, these data are limited. The EXPLORE study aimed to assess the incidence and indicators of suboptimal response to first-line anti-TNF agents in IBD patients in real-world practice in the newly industrialised countries.
Methods
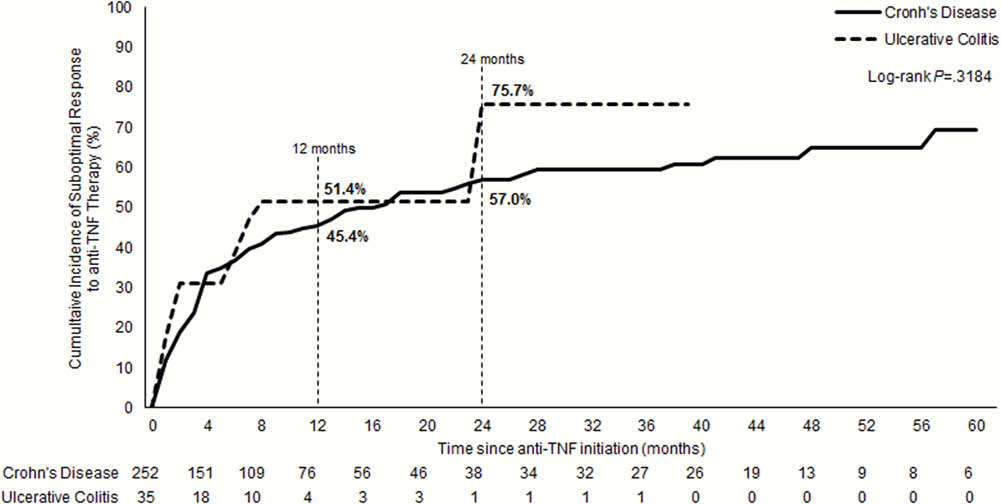
The EXPLORE study was a multinational, retrospective chart review study. In the China subgroup, adult patients from 10 centres diagnosed with ulcerative colitis (UC) or Crohn’s disease (CD) and who initiated first anti-TNF treatment between March 2010 and March 2015, were included. The cumulative incidence (CI) of suboptimal response was assessed over 24 months since the anti-TNF initiation. The indicators of suboptimal response included: IBD-related hospitalisation, dose-escalation, discontinuation including switch to another anti-TNF, non-biologic therapy augmentation, or IBD-related surgery. Primary non-response (PNR) and secondary loss of response (SLOR) were defined as any suboptimal response indicator at <4 and ≥4 months after anti-TNF initiation, respectively.
Results
Overall, 287 first-line anti-TNF treated patients (35 UC; 252 CD) were included: male, UC 54.3% (
Conclusion
Approximately three-quarters of UC and over half of CD patients experienced a suboptimal response to their first anti-TNF agent at 24 months. Given the high unmet needs observed with anti-TNF therapies in China, IBD patients may benefit from greater optimisation of care and new biologic options.


