P642 Serum adalimumab levels measured between days 9 and 13 from drug injection can be interpreted clinically in a similar way to trough levels in patients with inflammatory bowel disease
J. Guardiola Capón1, K. Serra1, L. Rodríguez-Alonso1, E. Santacana2, N. Padullés2, A. Ruiz-Cerulla1, C. Arajol1, B. Camps1, G. Surís1, E. Sanchez1, F. Rodríguez-Moranta1
1Hospital Universitari de Bellvitge, Gastroenterology, Barcelona, Spain, 2Hospital Universitari de Bellvitge, Pharmacy, Barcelona, Spain
Background
AntiTNF therapeutic drug monitoring is currently performed at trough, immediately before drug administration. However, in clinical practice when subcutaneous medications are used, blood extractions often do not coincide with that moment. The aim of this study was to know if adalimumab levels measured between injections are sufficiently similar to trough levels to be used in clinical practice in a similar way.
Methods
295 adalimumab level determinations performed at different time points of 99 injection cycles in 55 patients with inflammatory bowel disease (IBD) were included in the study. 51 patients received 40mg every 2 weeks and 4 patients received 80mg every 2 weeks.
Results
Median adalimumab levels (IQR) at trough, between days 1–4, 5–8 and 9–13 were 10.6 (6–12), 12.3 (7–18), 13 (7–19) and 10.8 (8–12), respectively. The median differences between trough level and days 1–4, 5–8 and 9–13 were 1.7 (IC 95% 1–2.3) (
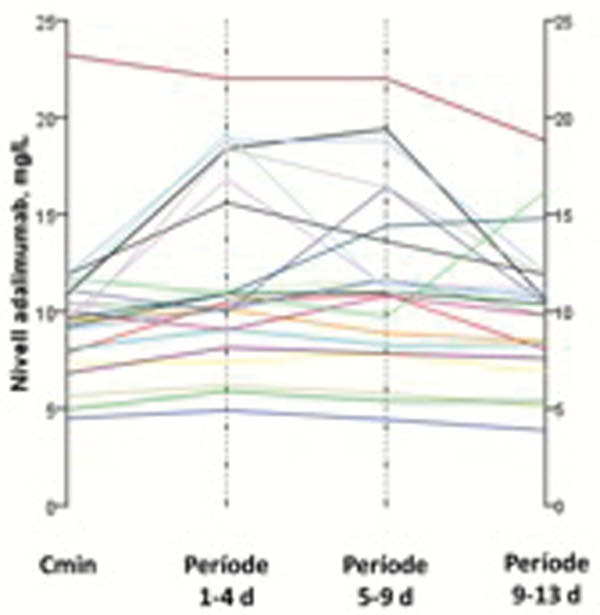
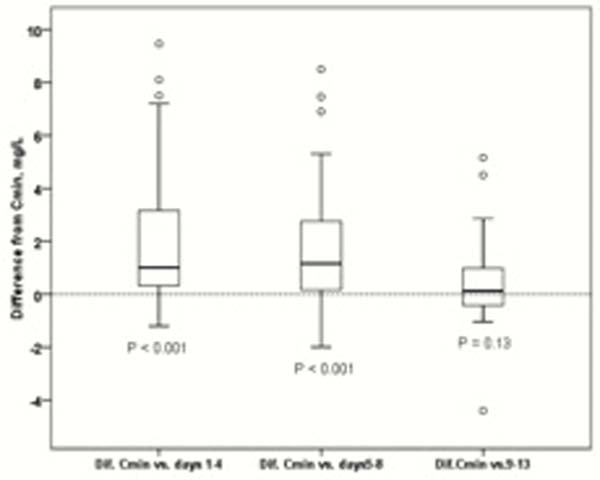
Conclusion
Adalimumab levels between days 9 and 13 from drug injection are very similar to trough level and could be interpreted clinically at the same way. Adalimumab levels between days 1 and 8 are significantly higher, although, differences are small.


