P823 DNA methylation profiles accurately predict vedolizumab response and remain stable during induction and maintenance treatment in Crohn’s disease
V. Joustra1, I. Hageman2, A. Li Yim3, K. Gecse1, M. Lowenberg1, A. te Velde2, J. Satsangi4, W. de Jonge2, P. Henneman3, G. D’Haens1
1Amsterdam UMC location AMC, Gastroenterology and Hepatology, Amsterdam, The Netherlands, 2Tytgat Institute for Liver and Intestinal Research- Amsterdam UMC, Gastroenterology, Amsterdam, The Netherlands, 3Genome Diagnostics Laboratory- Amsterdam UMC location AMC, Clinical Genetics, Amsterdam, The Netherlands, 4John Radcliffe Hospital, Translational Gastroenterology Unit, Oxford, UK
Background
While the use of biologicals has revolutionised the treatment of Crohn’s disease (CD) in recent decades, many patients fail to respond or lose response over time. Predictors for treatment selection and/or success are lacking. Vedolizumab (VDZ) induces corticosteroid-free clinical remission in 29% and endoscopic remission in 36% of patients after 6 months. Accumulating evidence suggests that the epigenome, mainly DNA-methylation, is associated with certain CD phenotypes. Here, we investigated whether the DNA methylome of peripheral blood (PB) obtained from CD patients treated with VDZ was associated with clinical and endoscopic response or non-response to treatment.
Methods
We prospectively recruited CD patients scheduled to start VDZ treatment at regular dosage and who underwent baseline endoscopy. PB was collected prior to treatment (T1) and at a median of 14 weeks (T2) into treatment. Additionally, a third PB sample was collected after a median of 79 weeks (T3) into treatment for 10 CD patients. DNA methylation was measured using the Illumina Infinium Methylation EPIC BeadChip. After a median 27 weeks of treatment, patients were characterised as responders (R) or non-responders (NR) based on endoscopic changes (R= ≥50% reduction in SES-CD score) combined with either steroid-free clinical remission (≥3 point drop in HBI, no systemic steroids) and/or biochemical response (≥50% reduction in C-reactive protein and faecal calprotectin).
Results
We analysed data of 16 R and 14 NR patients, 53% of which were female with a median age of 37. No significant differences in age, sex or smoking were observed between R and NR. All patients had measurable serum VDZ concentration at the second endoscopy. To identify response-associated biomarkers, we performed elastic net regression through repeated cross-validation. We identified 38 differentially methylated probes (DMPs) associated with response (Figure a), 7 of which were located in gene promoters. Performance-wise the DMPs displayed a sensitivity of 0.86 and a specificity of 0.8 in predicting response (Figure b). Notably, the degree of methylation for the response-associated DMPs did not change after 14 weeks of treatment (
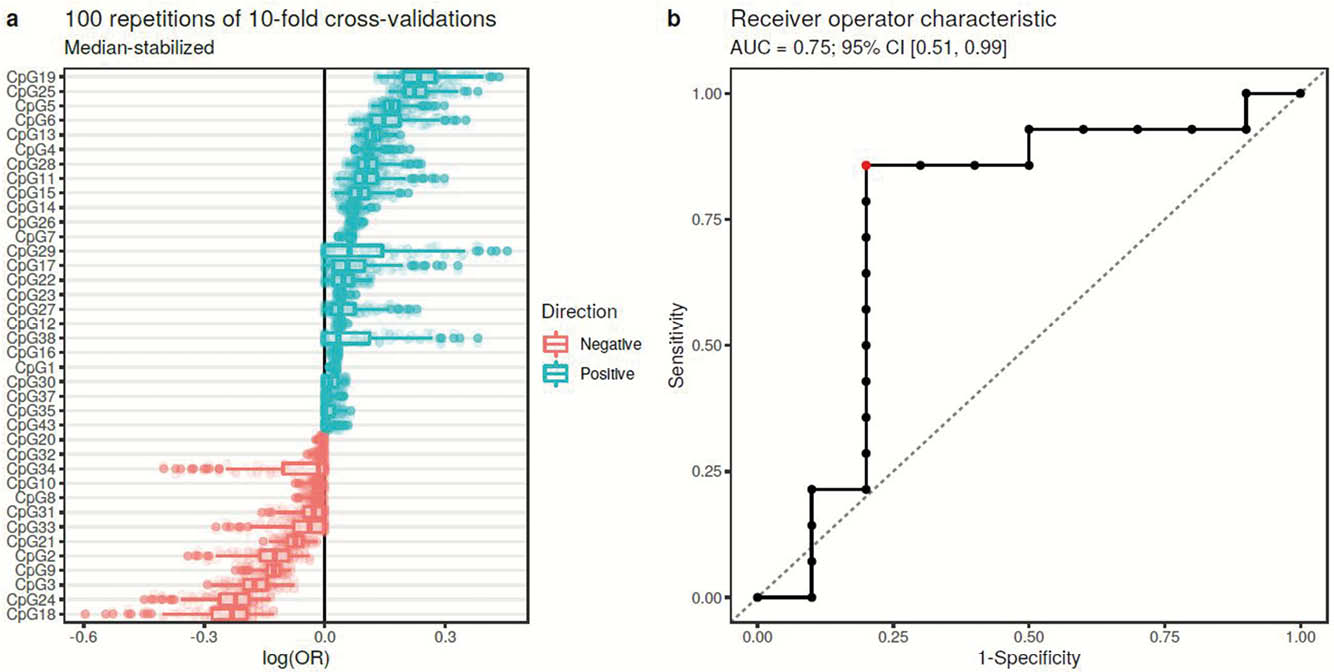
Conclusion
Here, we show that the DNA methylome in peripheral blood is associated with response to VDZ. As the difference in methylation remained stable during induction and maintenance treatment with VDZ, inflammation did not appear to affect the response-associated DMPs. Our observations provide evidence towards the establishment of epigenetic marks with predictive biomarker capabilities, paving the road towards personalised treatment selection in CD.


