P827 SARS-CoV-2 breakthrough infections after COVID-19 vaccination in patients with Inflammatory Bowel Disease: A systematic review and meta-analysis
Van De Pol, N.(1)*;Pan, Q.(1);Derikx, L.A.A.P.(1);Bakker, L.(1);van der Woude, C.J.(1);de Vries, A.C.(1);
(1)Erasmus MC, Gastroenterology and hepatology, Rotterdam, The Netherlands;
Background
Patients with inflammatory bowel disease (IBD) have reduced seroconversion rates to COVID-19 vaccination. It is unclear whether an impaired immune response in vaccinated IBD patients impacts the susceptibility to SARS-CoV-2 infection and occurrence of (severe) COVID-19. We evaluated SARS-CoV-2 breakthrough infection rates and the disease course of COVID-19 in vaccinated IBD patients.
Methods
A systematic literature search was performed for studies which reported SARS-CoV-2 breakthrough infection rates and/or the disease course of COVID-19 in patients with IBD after COVID-19 vaccination. Primary outcome was the rates of breakthrough infection per time period. In meta-analyses, the pooled relative risk was calculated with a random effects model for vaccinated patients compared to vaccinated controls, to partially vaccinated and unvaccinated patients with IBD.
Results
A total of 16 studies were included in analysis. The study period ranged from January 2020 to October 2021, and a follow-up time ranges from 3 weeks to 6 months. The breakthrough infection rates range from 0 to 37.4% in IBD patients within the study follow-up time. Strikingly, only studies with vaccination prior to December 2021 showed a breakthrough infection rate above 2%. (Figure 1). The disease course of a breakthrough infection is generally mild, with mild constitutional and respiratory symptoms in 85% of infected IBD patients. Hospitalization and mortality rates are low (0-8.7% and 0-4.3% respectively). Meta-analyses showed a significantly lower pooled relative risk of breakthrough infection for vaccinated as compared to unvaccinated IBD patients (RR 0.07, 95% CI 0.03; 0.18). No difference was observed in risk of breakthrough infections between IBD patients and non-IBD controls (RR 1.01, 95% CI 0.92; 1.10), and no difference between vaccinated and partially vaccinated IBD patients (RR 0.67, 95% CI 0.38; 1.18). The impact of immunosuppressive therapy on breakthrough infection rates differs between studies. One study reported higher breakthrough infection rates for patients treated with infliximab in comparison to vedolizumab (P<.05). Other studies showed no impact on the breakthrough infection rates for immunosuppressive treatment vs no treatment, anti-TNF-α/corticosteroids vs without anti-TNF-α/corticosteroids and other biologics vs anti-TNF-α.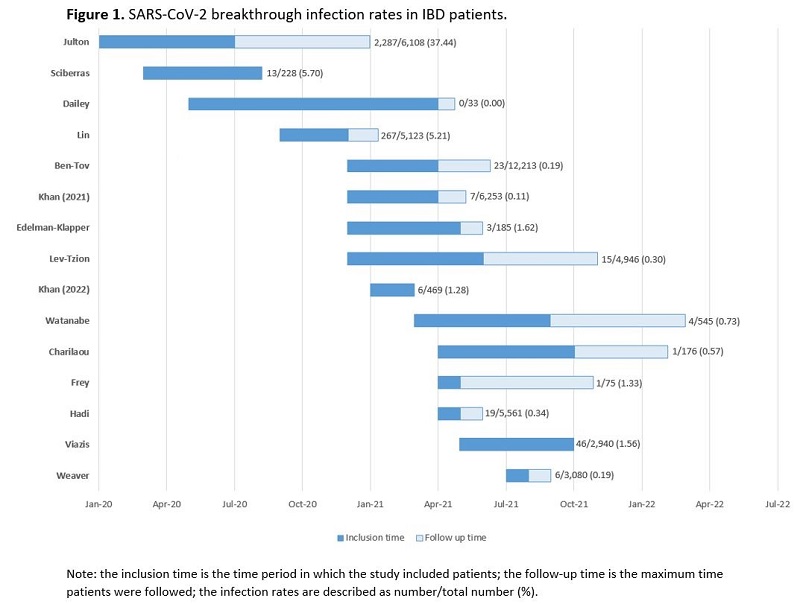
Conclusion
Vaccination is effective to prevent COVID-19 infections in patients with IBD. Breakthrough infections do occur, but the disease course is generally mild. Available data seem to suggest a declining trend of breakthrough infections during calendar time. Data on the impact of IBD medication on the rate of breakthrough infections and disease course require further elucidation.
- Posted in: Poster Presentations: Epidemiology 2023


